Articles
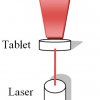
Counterfeiting is a major problem throughout the world and transmission Raman spectroscopy proves to be a very useful tool for fast and non-destructive detection of counterfeit pharmaceuticals.
The African crested rat chews poisonous bark and then applies its saliva to specialised hairs which provide a most effective protection. Maxime Boulet-Audet and Chris Holland describe this and the use of infrared spectroscopy to learn more about this unusual animal. The new knowledge may even lead to new treatments for human cardiac conditions.
Industrial environments pose potentially hazardous situations whereby workers may be exposed to various airborne toxic elements in their breathing zone. One of the main aerosol fractions of interest is welding fume, which can be determined with XRF spectrometry.
After an introduction to MALDI, DESI and SIMS, the authors describe their main applications in forensics, as well as the advantages provided in terms of sample preparation over approaches routinely used in toxicological laboratories.
Chemical contaminants in water are of concern to all of us. This article reviews the use of mass spectrometry (MS) in environmental and wastewater analysis.
Tea is harvested from the bud, the first leaf and the first three leaves up to a maximum of four leaves. Quality decreases from the bud down as the parts become older. This study looks at the use of NIR spectroscopy to determine optimal production using the harvested material.
TiO2 is widely used as a sunscreen UV filter and as a colouring agent in all types of cosmetic products. TiO2 has recently captured the attention of the scientific community since its safety assessment has been placed again under consideration. Inductively-coupled plasma atomic emission spectrometry (ICP-AES), square wave voltammetry (SWV) and sedimentation field-flow fractionation (SdFFF) are described in this article to characterise and quantify the TiO2 particles inside six commercial foundation creams.
Nuclear magnetic resonance (NMR) spectroscopy is one of the most powerful analytical tools used to probe details of molecular structure and dynamics. It requires very high magnetic fields and, hence, generally uses extremely large, powerful magnets. The advent of small, powerful magnets has allowed much less expensive low resolution NMR instrumentation to be designed, making it feasible to measure commercially important characteristics of dispersion behaviour and performance, including the wetted surface area of particulate suspensions and emulsion droplet size. An important additional practical application is the ability to determine competitive adsorption and/or displacement of polymers and surfactants at interfaces. This article presents a brief overview of these new approaches together with an example of each measurement.
This article shows that NIR spectroscopy offers the potential for bioprocess manufacturing companies to limit the variability in the biological production process, thereby increasing the yield and reducing the number of errors.
Applications of mass spectrometry in the clinical area have increased significantly in recent years and continue to expand. The complexity of analyses has also increased, with work being undertaken in clinical labs that would have been the reserve of research labs a few years ago.
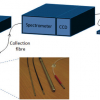
This article surveys developments in the applications of Raman spectroscopy and the design of probes for use in endoscopes for the detection of early cancers in the throat and stomach.
A poem with a spectroscopic theme by Professor Bill George of the University of Glamorgan.
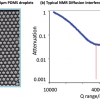
BBCEAS is a new cavity-based absorption technique that uses broadband sources rather than lasers. This offers potential improvements in signal-to-noise but often lower spectral resolution compared to techniques where the laser wavelength is scanned. The article concentrates on liquid-phase applications, an interesting new area since most cavity-based absorption studies have been performed on gas-phase species up to now.
As nanotechnology grows in importance, the need for rapid, easy and automated analytical methods to investigate new nanomaterials increases. Near infrared (NIR) spectroscopy has the potential to replace numerous methods for the investigation of a wide range of nanostructured materials and with particular potential in the area of PAT.
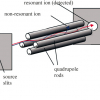
M. Hässig,a K. Altwegg,a H. Balsiger,a B. Schläppi,a J.J. Berthelier,b B. Fiethe,c S.A. Fuselier,d J. De Keysere and M. Rubinf
aPhysikalisches Institut, University of Bern, Sidlerstr. 5, CH-3012 Bern, Switzerland
bLATMOS, Saint Maur, France
cInstitut of Computer and Network Engineering, Technische Universität Braunschweig, Germany
The authors describe the use of a range of complementary methods to explore cellular, physiological and behavioural mechanisms underlying Al accumulation and toxicity, and its eventual fate, using the pond snail as a model organism.
Raman spectroscopy is to be used as one of the analytical techniques for the European Space Agency’s ExoMars mission to identify the geological and biogeological spectral signatures that could herald the presence of extinct or extant life on Mars. The article looks at the benefits of Raman spectroscopy for this and the research on Earth to build knowledge of the spectra of organisms living in extreme conditions here.
This article describes the analysis of cells from the pulp of the feather by IR spectroscopy and the possibility of using infrared imaging of intact eggs to determine bird gender.
Nati Salvadó, Salvador Butí and Trinitat Pradell have used a number of techniques to investigate changes in pictorial techniques in Catalan paintings in the 15th century. The combination of different techniques is of particular value. The use of synchtrotron radiation as a light source is also an advantage.
Roumiana Tsenkova introduces us to the field of “Aquaphotomics” which she has pioneered. The potential information held by water in biological systems coupled with NIR spectroscopy’s ability to extract a large amount of information at once, means that Aquaphotomics may be of great importance in the study of living organisms.