Jennifer A. Dougan and Sergei G. Kazarian
Department of Chemical Engineering, Imperial College London, SW7 2AZ, UK. E-mail: [email protected]
Introduction
The development of tools which enable the measurement of biological responses to external stimuli, in situ, unobtrusively and in a chemically informative way, is integral to our ability to deliver results for biomedical applications. Fourier transform infrared (FT-IR) and Raman spectroscopies are already making significant contributions in this field.1 Producing molecularly specific, information-rich spectra, these non-destructive techniques offer the opportunity to interpret bonding and environmental changes in biologically important situations. Using FT-IR spectroscopy, coupled with a focal plane array (FPA) detector, it is now possible to obtain chemical images of living cells.2,3 Here we highlight some initial developments in this field.
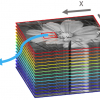
Historically, it was challenging to obtain meaningful information from FT-IR measurements in aqueous environments due to the strong absorption of water in the mid-IR region. Dominant absorption bands occur around 3600 cm–1 and 1640 cm–1 as a result of OH stretching and bending modes, respectively. Nevertheless, dried samples such as tissues and cells have been well characterised by FT-IR spectroscopy. In recent years, the challenge presented by aqueous environments has been mitigated by the use of attenuated total reflection (ATR) FT-IR spectroscopy2 or, in transmission measurement mode, by careful preparation of samples,3 or use of infrared radiation from a synchrotron source.4 The ability to measure FT-IR spectra in aqueous environments is important because it allows FT-IR imaging to be applied to live cells. Imaging, as opposed to mapping, records all spectra simultaneously and simple treatment of the acquired data produces “chemical photographs”; this is shown schematically in Figure 1.

FT-IR imaging
FT-IR imaging is made possible by coupling a spectrometer with a focal plane array (FPA) detector. Instead of a single spectrum being produced, the FPA detector—a grid of detectors (typically of 64 × 64 or 128 × 128 elements)—allows thousands of spectra to be concurrently recorded. Since a full spectrum is obtained at each point of the sample simultaneously, the resulting images are spectrally, spatially and temporally resolved. This has significant implications in the study of dynamic systems.
ATR FT-IR spectroscopy
Technical details and biological applications of ATR FT-IR spectroscopy have previously been described.5,6,7 Some features are described briefly here which explain the suitability of this approach to the analysis of aqueous biological samples. ATR FT-IR spectroscopy is made possible by the interaction between IR light and an absorbing sample at its interface with a high refractive index material. Incident light is shone through an IR transparent material of high refractive index, an internal reflection element [(IRE), for example, ATR crystal], above a critical angle. At the interface between the ATR crystal and a sample of lower refractive index, light is internally reflected producing an evanescent wave which penetrates into the sample. Absorption at discrete IR frequencies by the sample results in the attenuation of the incident IR light. Coupling this process with a single element infrared detector and spectrometer produces an ATR FT-IR spectrum.
The evanescent wave typically penetrates into the sample by only 0.2–5 µm (when defined by the depth of penetration). The exact value is dependent upon a number of parameters, such as the refractive indices of both materials, the wavelength of light and the angle of incidence. Nevertheless, the small depth of penetration results in a small effective pathlength. In practise, this means that samples can be analysed in the presence of a highly absorbing species such as water without loss of spectral integrity in the regions of interest.
ATR FT-IR imaging of live cells
Single reflection ATR FT-IR spectroscopic imaging can be carried out in a number of optical setups which have been reviewed elsewhere.5 These different approaches range from using diamond, Si or ZnSe, as the IRE in a large sample compartment to a Ge IRE coupled with an IR microscope. These options provide a large choice in both the field of view and spatial resolution. The best spatial resolution is obtained using micro ATR FT-IR imaging; i.e a Ge ATR objective coupled with an IR microscope. This approach has recently been applied to study the crystallisation of proteins.8 The field of view in micro ATR is ca 64 µm × 64 µm with a spatial resolution of ca 4 µm (in the mid-IR “fingerprint region”. For cell studies this is attractive as it allows sub-cellular components to be visualised. In some cases, though, it may be more relevant to study cell populations and their response to environmental stimuli. In such cases, macro ATR FT-IR, using diamond as the IRE, in a large sample compartment may be preferable. Here, the field of view is typically 500 µm × 700 µm with a spatial resolution of 15–20 µm. The fields of view and spatial resolution make ATR FT-IR imaging studies relevant for cell investigation and, as mentioned previously, the small depth of penetration of ATR FT-IR spectroscopy allows measurements to be taken in aqueous environments. It is essential to ensure that cells are in close contact with the ATR crystal. This was achieved by culturing adherent cancer cells directly onto the ATR crystal, Figure 2(a).2,6 A thin layer of poly-L-lysine was used to facilitate good adhesion between the cells and the ATR element surface. The micro ATR approach allows sub-cellular regions to be identified, see Figure 2(c). The amide II band at 1535 cm–1 typically indicates the presence of proteins; plotting the distribution of the integrated absorbance of that band allowed the location of cells on the crystal to be identified. Areas corresponding to cell nuclei were then observed by plotting the distribution of the integrated absorbance of the phosphate stretching band of DNA, 1084 cm–1. Glycogen-rich regions were identified by the 1023 cm–1 band. Since glycogen levels are known to be linked to abnormalities or disease, it is of interest to be able to identify this quantitatively at the single cell or cell population level. Macro imaging was carried out2 in order to observe the response of a cell population to starvation, see Figure 3. The cells were cultured and allowed to adhere to the diamond ATR crystal. Swapping the media for phosphate buffered saline (which does not provide nutrients for cell growth or sustenance) and monitoring the glycogen level as a function of time, allowed cell starvation to be observed by FT-IR imaging. The amide II and DNA bands are still observed after 90 min, indicating that the cells remain attached to the surface, whereas there is a marked decrease in the integrated absorbance of the glycogen band. In contrast, after 3 h of medium replacement, there is a reduction in all bands as cells detach from the ATR crystal surface in response to starvation.2


Transmission mode FT-IR imaging of live cells
As mentioned, typically, transmission mode measurements of live cells have been limited to using a synchrotron IR source in order to overcome some of the difficulties associated with the strong IR absorbance of water obscuring bands of interest. Recently, live cells have been studied in transmission mode imaging using a conventional globar mid-IR source, see, for example, Figure 4.3 A microfluidic device was prepared between thick IR transparent windows (CaF2), to which cell culture medium was introduced. The cells adhered to the bottom window before the device was sealed to 10 µm thickness by use of a spacer. Imaging measurements through thick infrared transparent windows suffer from chromatic aberration resulting in a wavelength-dependent spread of focus in the z-direction. The use of two CaF2 lenses, carefully positioned, above and below the windows eliminates this chromatic aberration by allowing all wavelengths to focus at the same point, Figure 4(a).3 By using this approach, it was possible to clearly image cell position according to the amide II band, Figure 4(b). There was a magnifying effect of the pseudo-sphere arrangement which allowed sub-cellular differences to be resolved. Extracted average spectra from the cell indicated a change in DNA/RNA conformation upon cell spreading, Figure 4(c). The transmission measurement mode methodology was further extended to study live cells in droplets of media which were created in oil flow.3 Individual droplets were produced in a microfluidics device which contained one or two cells per droplet. Although the cells could not be imaged during flow, once stationary, the imaging of single cells could be achieved, opening up the possibility for single cell studies in discrete environments with chemical imaging.

Conclusions
Live cell spectroscopic imaging studies using a standard globar IR light source show promising results in both the ATR and transmission measurement modes. This is an exciting area of biomedical research with opportunities to examine the influence of various environmental stimuli. By combining FT-IR imaging methodology with microfluidics devices, the opportunity to study live cells by FT-IR imaging in controlled environments is now possible and further work in this area is underway in the authors’ laboratory.
References
- R. Salzer and H.W. Siesler, Eds, Infrared and Raman Spectroscopic Imaging, 1st Edn. John Wiley & Sons Ltd, Chichester, UK (2009).
- M.K. Kuimova, K.L.A. Chan and S.G. Kazarian, “Chemical imaging of live cancer cells in the natural aqueous environment”, Appl. Spectrosc. 63(2), 164 (2009). doi: 10.1366/000370209787391969
- K.L.A. Chan and S.G. Kazarian, “Aberration-free FTIR spectroscopic imaging of live cells in microfluidic devices”, Analyst 138(14), 4040 (2013). doi: 10.1039/C3AN00327B
- C.J. Hirschmugl and K.M. Gough, “Fourier transform infrared spectrochemical imaging: review of design and applications with a focal plane array and multiple beam synchrotron radiation source”, Appl. Spectrosc. 66(5), 475 (2012). doi: 10.1366/12-06629
- S.G. Kazarian and K.L.A. Chan, “Micro- and macro-attenuated total reflection Fourier transform infrared spectroscopic imaging”, Appl. Spectrosc. 64(5), 135A (2010). doi: 10.1366/000370210791211673
- S.G. Kazarian and K.L.A. Chan, “ATR-FTIR spectroscopic imaging: recent advances and applications to biological systems”, Analyst 138(7), 1940 (2013). doi: 10.1039/c3an36865c
- S.G. Kazarian and A.V. Ewing, “Applications of Fourier transform infrared spectroscopic imaging to tablet dissolution and drug release”, Expert Opin. Drug Del. 10(9), 1207 (2013).doi: 10.1517/17425247.2013.801452
- S.E. Glassford, L. Govada, N.E. Chayen, B. Byrne and S.G. Kazarian, “Micro ATR FT-IR imaging of hanging drop protein crystallisation”, Vib. Spectrosc. 63, 492 (2012).doi: 10.1016/j.vibspec.2012.07.011