Peter J. Jenks, BSc, MRSC
The Jenks Partnership, Newhaven House, Junction Road, Alderbury, Salisbury, Wiltshire SP5 3AZ, UK
The first article I wrote for Spectroscopy Europe was in Vol. 13 No. 6, published in December 2001. Together with my co-author Dr Henryka Jerzak, I described the process then followed by the Polish Institute for Industrial Organic Chemistry to certify pure pesticides for purity. We described how GC-FID, GC-MS, HPLC, TLC and DSC are all used together to try and produce a reliable estimate of purity, but in our conclusions we were left asking the following questions:
“What does purity mean in terms of complex organic molecules?”
“How should the purity of such materials be really examined?”
“What is the real meaning of traceability to the mole in the commercial analytical world?”
In the last 11 years there have been many developments in analytical chemistry, but for the most part the certification of pure organic molecules, now mostly in laboratories accredited to IS017025 + ISO Guide 34, proceeds much as then. Instrumentation has improved, data can be developed with smaller analytical uncertainties, but the result is still the development of a mass of data from which it is possible to estimate the levels of impurities, moisture and the molecule in question and so derive a purity value that forms the basis of the Certificate of Analysis. But this is essentially an indirect procedure which results in a “best estimate” of the purity, and its associated uncertainty.
Earlier this year, talking to scientists at both LGC and at Sigma Aldrich, I became aware of the potential for quantitative nuclear magnetic resonance or “qNMR” to short cut all this work and allow the direct certification of almost any organic molecule. How can this be?
Nuclear magnetic resonance, or NMR as it is generally known, is not a new technique. Although NMR was first described in 1938 by Isidor Rabi,1 it was only in 1946 when Felix Bloch and Edward Mills Purcell developed NMR to work on liquids and solids that the true analytical potential of NMR was appreciated. NMR was a spin-off from the rapid development of RADAR during WWII. All the early pioneers noticed that magnetic nuclei, such as 1H and 31P, could absorb RF energy if placed in a magnetic field when the RF was of a frequency specific to the identity of the nuclei. When this absorption occurred the nucleus was described as being “in resonance”. It was soon realised that different atomic nuclei within a molecule resonate at different (radio) frequencies for the same magnetic field strength. So the resonance frequency of a particular substance is directly proportional to the strength of the applied magnetic field. This means that NMR has the potential to be a powerful spectroscopic tool.
Since 1946 NMR has been much developed, but unlike the other modern analytical technique mass spectrometry, or MS, it has not become a common analytical tool, rather it became widely used in structural analysis—the majority of NMR applications have been qualitative.
In the early 1970s, Joseph Ray looked at quantitative NMR, when he was working for the Standard Oil Company (later Amoco and then BP). Ray was using a Varian CFT-20 spectrometer that was dedicated to 13C observation at 20 MHz. It became clear that quantitative NMR (qNMR) represented a metrological method of analysis with unique capabilities but limited practicality, since the time required for and throughput of routine analyses were impossibly slow.
Since the 1970s developments by manufacturers such as Bruker, Jeol and Varian including advances in the frequency and power of the magnetic field, from about 20 Mz then to close to 1 GHz now, and in associated software, both for the control of the machine and the analysis of data, mean that qNMR has become a viable analytical technique.
One of the original versions of NMR, 1H NMR (HNMR) which focuses on the hydrogen-1 nuclei within the molecules of a substance, has become the analytical method of choice for qNMR. Initially used to determine the structure of molecules, quantitative 1H NMR or qHNMR has been gaining popularity in the analysis of complex natural products such as botanicals and dietary supplements. This is because peak areas are proportional to the number of corresponding nuclei and most importantly the signal intensity is independent of the chemical nature of the substance, so there is no need to chemically identify the impurities in a material under examination.
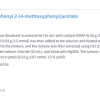
From the point of view of the certification of an organic material, it is this directness that is the most important feature of qNMR. The time consuming estimation of the amount of impurities and solvents can be ignored: all that is needed is a direct measurement of the analyte. This is achieved by comparing the signal intensity from a primary certified reference material and the unknown, under the same analytical conditions. For example, to determine the purity of the amino acid l-serine, potassium hydrogen phthalate (NIST SRM84k, 99.9911% w/w) can be used as internal standard. To cover all measurement challenges associated with organic molecules a series of secondary qNMR internal standards needs to be developed from two or three primary internal standards. This family of calibrators must be compatible with all expected NMR solvents, fit the molar mass/H ratio and chemical shift of the target molecule.
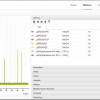
Figure 1 shows how qNMR is used to develop a certified reference material. There is, unfortunately, one reason why qNMR is unlikely to be adopted by the many producers of certified reference materials: cost.

To achieve satisfactory resolution and response time, modern 600 Hz NMR is probably optimal. Such NMR machines are expensive: adding the associated auto samplers and sample preparation equipment means that an investment of more than E1 million is required. Yes, there will be some producers who take the step, but unless and until the price of such NMR machines falls, the technique is unlikely to become mainstream. This is unfortunate as qNMR could be attractive as an analytical instrument, replacing GC-MS and LC-MS in many areas.
Why should the development of affordable routine qNMR be important? It is the directness of the technique that is the key. All existing methods require the availability of calibration CRMs. qNMR does not.
Supply of calibration standards is not seen as a problem and qNMR will improve the availability of CRMs to be used for calibration. But as REACH develops and extends its scope in a number of areas, controls or prohibitions on the supply and use of highly toxic chemicals are likely to compromise the availability of the CRMs that are required to calibrate traditional analytical systems. Many of the molecules likely to be restricted, or banned, are cited in legislation that requires their measurement in materials critical to human health. But if the instrument systems cannot be calibrated by traditional means, how can the legislated analysis be completed?
Does qNMR offer an answer?
Can affordable qNMR be foreseen?
I believe so: 30 years ago mass spectrometry was an analytical technology that was seen as way out of the reach of the routine analytical laboratory. Today MS detectors are almost commonplace, and so-called “hyphenated systems” are in almost every analytical laboratory. The rate of development of analytical instruments seems to accelerate, perhaps it will be only 5–10 years before we see qNMR displacing GC-MS and LC-MS in routine environmental, food and pharmaceutical laboratories.
Reference
- I.I. Rabi, J.R. Zacharias, S. Millman and P. Kusch, “A new method of measuring nuclear magnetic moment”, Phys. Rev. 53(4), 318–327(1938). doi: 10.1103/PhysRev.53.318