SCIEX is to collaborate with the laboratory of Amanda Paulovich at Fred Hutchinson Cancer Research Center, Seattle, WA, USA, to make Targeted Proteomics in cancer research more reproducible and specific. The collaboration provides SCIEX rights to commercialise the immuno-MRM assays that have been developed in the Paulovich Laboratory, a member of the US National Cancer Institute’s Clinical Proteomic Tumor Analysis Consortium. This effort, aligned with the National Institute of Health’s (NIH) strategy to make technology more widely accessible through public/private partnerships, will result in commercially available assays that quantitatively measure phosphorylated and unmodified proteins known to be involved in cancer signalling pathways.
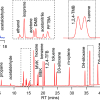
Targeted Proteomics was named “Method of the Year” by Nature Methods in 2013, and Dr Christie Hunter at SCIEX was awarded the Human Proteome Organization’s 2013 Science and Technology Award for her contributions to the development and commercialisation of this technology. To extend the reach of this technology, and make it more sensitive, and more routine and reproducible, an augmented approach is required over direct-MRM. Immuno-MRM assays combine the best features of immunoassays and mass spectrometry to provide highly reproducible, specific and sensitive quantification of target proteins, including phosphorylated proteins. However, a lack of availability of off-the-shelf content for this technique has been holding the field back, and this partnership aims to redress that.
By partnering with the Paulovich Laboratory, SCIEX will offer researchers a complete solution for mass spectrometry-based protein quantification of specific key biological pathways. At the HUPO 2015 Annual World Congress, SCIEX introduced an improved targeted proteomics workflow that includes the new QTRAP 6500+ system and microflow LC, to combine high sensitivity and high throughput, and the Beckman Biomek Laboratory Automated Workstation, with optimised workflows for protein digestion. The immuno-MRM kits will be commercialised for this workflow, creating a solution that will include sample preparation reagents, antibodies and beads for target enrichment, internal standards for quantification, and related methods for LC-MS and data analysis.
This collaboration will produce highly validated off-the-shelf assays for running immuno-MRM experiments, which will improve biological reproducibility and thereby drive actionable research, which can be shared across labs. The immuno-MRM kits will be available in 2016.