Spotlight on nuclear magnetic resonance: a timeless technique
Clemens Anklin
Bruker BioSpin Corp., 15 Fortune Drive, Billerica, MA 01821-3991, USA. E-mail: [email protected]
Introduction
Nuclear magnetic resonance (NMR) spectroscopy has evolved immensely since its early use solely in physics. Since then, chemists have come to rely on NMR for determining molecular structure, and scientists now use this method for a broad range of applications in a diverse range of fields. For example, a recent poll of methods utilised by pharma discovery scientists searching for the next new blockbuster drug highlights the dominance of NMR spectroscopy in fragment-based lead generation.1 In addition, workers investigating food fraud used NMR to identify imitation cheese and ice cream—products in which the milk fat and/or milk protein had been replaced with cheaper, non-milk components, such as soy, starch or vegetable oil.2
This article looks back at the work of NMR’s founding scientists and reflects on key developments that have propelled NMR to its position today—as the go-to technique for scientists looking for an information-rich and non-destructive analytical tool to reveal the structure, identity, concentration and behaviour of molecules in solid or liquid samples.
In the beginning
The earliest NMR pioneers developed and built their own instrumentation. As the technology advanced, few chemists were prepared to invest the time and money required to build their own spectrometers, so the full impact of NMR as a new analytical technique was not realised until they could be purchased from commercial companies. Such early companies included Varian Associates (CA, USA), Japan Electron Optics Ltd, Associated Electrical Industries (UK), Perkin Elmer Corporation (UK) and Trüb Täuber (Switzerland, later Bruker-Spectrospin). These companies did not stand to make much money from NMR, as the market was so limited at the time. The definitive commercial breakthrough came with the introduction of the Varian A60, which paved the way for high-resolution NMR in organic chemistry.
Although there are small number of key players in the modern NMR market, such as JEOL, Nanalysis, Oxford Instruments and Thermo Fisher, NMR is indivisible from the early development of the two key companies that went on to establish the field—Bruker and Varian. Günther Laukien, founder of Bruker, dedicated his post-doctoral studies to NMR spectroscopy and, in 1958, published his pioneering paper on high-frequency NMR.3 This described the theoretical aspects of what was known at the time, while also covering the practical considerations of constructing experimental systems.
Varian was established in 1948 within the Stanford Industrial Park by scientists from Stanford University. One of the early goals of the company was to commercialise the co-discovery of NMR spectroscopy by Felix Bloch in 1946. Subsequently, Edward M. Purcell and Felix Bloch were honoured with the Nobel Prize for Physics in 1952 for this work.
At around the same time that Laukien was studying, Varian had begun building the first commercial high-resolution spectrometers. They were based on continuous-wave sweep methods and electromagnets, and designed for use in analytical chemistry. When Laukien founded Bruker Physik AG in 1960, its earliest NMR spectrometer’s application was restricted to physical research laboratories, rather than for routine chemistry applications like Varian’s. Laukien’s acquisition of the Trüb–Täuber company (to become Spectrospin AG) enabled the newly-formed Bruker-Spectrospin to extend to high-resolution NMR. This, in combination with Bruker-Spectrospin harnessing the Fourier transformation (FT) method with the first FT-NMR spectrometer, eventually set the company up to overtake Varian as market leader. A possible oversight from Varian at the time as to the significance of FT-NMR, possibly due to the original founder’s replacement by sales-driven individuals without a technical background, allowed Bruker to overtake.4
The intense competition between these two companies drove many of the early developments and innovations in NMR for the next 20–30 years. In addition, the inputs from collaborations with several key scientists were crucial to the technological development and commercial success of these companies. For example, Dr Werner Tschopp and Dr Tony Keller contributed immensely to Bruker’s success as a result of the acquisition of Spectrospin. Innovations from Keller led to the first commercial implementation of the Fourier concept at Bruker. See Table 1 for a summary of some key milestones.
Table 1. Early NMR development.
1937 | Nuclear spin first measured in by Isidor Isaac Rabi in lithium isotopes and protons. |
1943 | Nobel Prize for Physics awarded to Rabi for this work. |
1946 | Discovery of nuclear magnetic resonance spectroscopy by Edward M. Purcell and Felix Bloch. |
1948 | Varian established in USA. |
1956 | Nobel Prize for Physics awarded jointly to Edward M. Purcell and Felix Bloch for their 1946 discovery. |
1961 | Introduction of the first high resolution NMR spectrometer—the Varian A60. |
1960–65 | Professor Günther Laukien founded Bruker and the development of its first NMR spectrometers began with the production of laboratory magnets and power supplies. Establishment of Spectrospin AG, a company initially founded as Trüb & Täuber in Zurich before being acquired by Bruker. First NMR, KIS (nuclear induction spectrometer), operated at 25 MHz using a permanent magnet. KIS 2 introduced for high-resolution spectroscopy (90 MHz). |
1964–66 | Ground-breaking research on the use of Fourier transform algorithm by Ernst and Anderson published. FT-NMR first described. |
1967 | Culmination of Bruker/Spectrospin developments—introduction of first fully transistorised NMR instrument, the Bruker HFX 90.
|
1969 | The world’s first commercial FT-NMR spectrometer system introduced by Bruker.
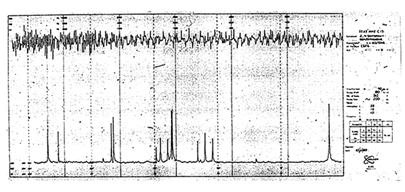
Carbon FT-NMR spectrum presented by Dr Tony Keller in 1969 at the Pacific Conference on Chemistry and Spectroscopy in Anaheim. |
1970–75 | The work on FT-NMR by Ernst and Anderson in 1966 required minicomputers to generate tapes that could be then be processed on larger computers. The advent of smaller, cheaper, and faster computers in the early 1970s made FT-NMR all but ubiquitous. |
From expert to everyday
After around 30 years of development, NMR was a maturing technique and was widely applied; there was hardly a paper in organic chemistry that did not report NMR data. When considering the developments from those initial NMR systems, three key areas can be identified that led to the systems today: larger magnets to improve sensitivity; improved probe technology and new designs to enhance performance; and a rapid increase in computer power to enable software to be developed that simplifies data processing and opens up the technique to non-experts. Table 2 highlights some key milestones in this “second-wave” of development. Interestingly, several of the pioneers and early adopters of NMR have published personal accounts and reviews of the development of the technique through the decades from 1980 to 2010.4–6
Table 2. Key developments in NMR from the 1980s to the present: decade by decade.
1980s | 2D NMR development comprised a huge step in the collection and analysis of data. Structure analysis of small and large molecules became possible and proved very useful in molecular biology and chemistry. These systems provided a tool that an everyday chemist could use. |
1990s | Higher dimensional spectroscopy (3D) developed. This was made possible by rapid developments in computer technology. The Fourier transformation of three and even higher dimensional NMR data can now be performed in a matter of seconds. Much larger protein molecules can now be analysed. Cryogenic probes first developed—significant reduction in noise from random thermal motion and this, together with tuned electronics, delivers around a five-fold enhancement in signal-to-noise ratio compared to an equivalent room temperature probe. In 1991, Richard Ernst was honoured with the Nobel Prize for Chemistry for his contributions to the development of the methodology of high-resolution NMR spectroscopy—work that begun with his Fourier transform research in the mid-1960s. |
2000s | Developments in cryogenic probes further improve performance, ease of use and system automation. In 2002, Kurt Wüthrich won the Nobel Prize in Chemistry, for his development of NMR spectroscopy for determining the three-dimensional structure of biological macromolecules in solution. |
2010s | Bruker introduces world’s first commercially available solid-state dynamic nuclear polarisation-enhanced NMR system (DNP-NMR). Microwave irradiation is used to transfer polarisation from unpaired electron spins to nuclear spins. As a result, polarisation enhancement yields a factor of up to 200 gain in sensitivity for solid-state NMR. Varian acquired by Agilent in 2010. Agilent exit the NMR business in 2014. |
Pushing the boundaries: current trends
The reach of NMR continues outside its legacy applications. Solid-state NMR has developed rapidly in the past decade, particularly in the field of materials science. The technique can be used to solve structural questions for batteries, polymers, crystalline solids and amorphous solids, particularly in disordered materials. Battery science, for example, is the subject of growing research interest due to the focus on electric cars and renewable energy storage. Solid-state NMR can provide insights into the structure, local order, molecular mobility and chemical processes in batteries, allowing researchers to study the composition and chemical reactions during charging and discharging, as well as electrical and magnetic properties. Recent developments in high magnetic fields and probes make low gamma nuclei much more accessible with solid-state NMR, while recent developments in very fast magic angle sample spinning enable the study of highly anisotropic systems, such as the paramagnetic materials often used in batteries.
Although an extremely useful technique, NMR is not as sensitive as other methods. The sensitivity of solid-state NMR for material science, pharmaceutical and biological solids applications can be significantly enhanced, however, using a dynamic nuclear polarisation-enhanced NMR system (DNP-NMR). DNP experiments transfer polarisation from electron spins to nuclear spins, for large signal enhancements and reduced signal averaging time. There is a huge capacity for DNP-NMR to become a routine method.
Another significant change in the application of NMR spectroscopy is in metabolomics. By collecting large amounts of spectra and gathering underlying data on metabolites, statistical analysis can reveal markers for a certain disorder or disease. Once a model has been established, the measurement of a single sample allows the classification of that sample as normal or abnormal—and even allows a diagnosis as to the nature of a disease. This takes NMR and places it in the hands of clinical scientists. They can ask: is this what I expected? And answer questions with “yes” or “no”.
In biopharma, researchers are now using NMR for the characterisation of the structure of monoclonal antibodies (mAbs). Another application of NMR in scale-up or production of biologics is the monitoring of the composition of growth media. Recognising the depletion of certain nutrients or the accumulation of potentially toxic metabolites can significantly improve yield as well as the efficiency of the fermentation.
The use of fluorine in the pharmaceutical industry has dramatically increased over the past few years. Today, five of the top ten selling small molecule drugs contain fluorine. 19F NMR offers a unique method in drug discovery, but also in the characterisation and quantification of fluorine-containing molecules. This has driven the launch of several cryoprobes that are capable of observing this nucleus with very high sensitivity.
Conclusion
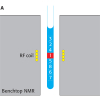
More than 70 years ago, a talented scientist first measured nuclear magnetic spin, work that resulted in the award of a Nobel Prize in 1943. Over the period from the mid-1950s to 2010, a small group of companies drove the development of increasingly sophisticated NMR spectrometers for a growing number of applications. Traditionally, larger instrumentation conferred higher quality results, but a new wave of benchtop NMR systems have proven their use in a range of laboratories, as well as in quality control and teaching applications, and in clinical diagnostics. A select group of companies, including Magritek, Nanalysis and T2 Biosystems, are developing ever more compact systems (the smallest being handheld devices), separate from the manufacturers of larger instruments.
There are a number of reasons why NMR is the method of choice for a broad range of analytical laboratories: it provides a non-destructive view of molecular dynamics in the solid state or solution, leaving samples intact for further analysis. The abundance of established methods and diversity of instrument size and sensitivity enables almost any analytical laboratory to utilise NMR for characterising molecular structures, monitoring the composition of mixtures, studying molecular dynamics and interactions, and quantifying known and unknown components.
References
- http://practicalfragments.blogspot.ch/2016/10/poll-results-affiliation-metrics-and.html
- Y.B. Monakhova, R. Godelmann, C. Andlauer, T. Kuballa and D.W. Lachenmeier, “Identification of imitation cheese and imitation ice cream based on vegetable fat using NMR spectroscopy and chemometrics”, Int. J. Food Sci. 367841 (2013). doi: https://doi.org/10.1155/2013/367841
- G. Laukien, Handbuch der Physik—Encyclopedia of Physics, Vol. 38/1, Ed by S. Flügge. Springer, Berlin, pp. 120–376 (1958).
- R.R. Ernst, “Zurich’s contributions to 50 years development of Bruker”, Angew. Chem. Int. Ed. 49, 8310–8315 (2010). doi: https://doi.org/10.1002/anie.201005067
- M. Antalek, https://benchtopthoughts.com/2014/10/17/the-end-of-an-era-varian-and-the-birth-and-growth-of-nmr/ (accessed 15 May 2017).
- J.W. Emsley and J. Feeney, “Forty years of Progress in Nuclear Magnetic Resonance Spectroscopy”, Progr. Nucl. Mag. Reson. Spectrosc. 50, 179–198 (2007). doi: https://doi.org/10.1016/j.pnmrs.2007.01.002