Mark J. Tobin,a* Ljiljana Puskar,b Song Ha Nguyen,c Jafar Hasan,d Hayden K. Webb,c Carol J. Hirschmugl,e Michael J. Nasse,f Gediminas Gervinskas,g Saulius Juodkazis,g Gregory S. Watson,h Jolanta A. Watson,h David E. Mainwaring,c Peter J. Mahon,c Richard Marchant,i Russell J. Crawfordc and Elena P. Ivanovac
aInfrared Microspectroscopy Beamline, Australian Synchrotron, 800 Blackburn Road, Clayton, Victoria, 3168, Australia
bMethods for Material Development, Helmholtz-Zentrum für Materialien und Energie GmbH, 12489 Berlin, Germany
cFaculty of Science, Engineering and Technology, Swinburne University of Technology, PO Box 218, Hawthorn, Victoria, 3122, Australia
dIndian Institute of Science, Bengaluru, Karnataka, India
eDepartment of Physics, University of Wisconsin–Milwaukee, 1900 E. Kenwood Road, Milwaukee, Wisconsin 53211, USA
fKarlsruhe Institute of Technology, Laboratory for Applications of Synchrotron Radiation, Kaiserstr. 12, 76131 Karlsruhe, Germany
gCentre for Micro-Photonics, Faculty of Science, Engineering and Technology, Swinburne University of Technology,
PO Box 218, Hawthorn, Victoria, 3122, Australia
hFaculty of Science, Health, Education and Engineering, University of the Sunshine Coast, Maroochydore DC, Queensland 4558, Australia
iMuseum Victoria, Melbourne, Victoria, 3001, Australia
Introduction
Insects and plants have evolved highly sophisticated surfaces that provide them with a number of functional advantages for dealing with their environment, and imparting beneficial properties to these organisms.1 For example, the surface of some leaves, such as those of the lotus plant, are well known to have highly water repellent, or superhydrophobic, properties2 and this provides the plant with self-cleaning leaf surfaces, benefiting the plant by inhibiting bacterial or fungal infection, or possibly ensuring optimum photosynthetic ability. This is sometimes referred to as the “lotus effect”. Similar superhydrophobic behaviour has been observed on insect cuticles, in particular the wings of certain groups such as cicadas, dragonflies and damselflies, where the water repelling properties enable the insects to remain clean and dry in wet and dirty environments.3,4,5 Indeed, some insect wing surfaces have recently been shown to have additional antibacterial properties.6 Such superhydrophobic natural surfaces have attracted the attention of materials scientists for their potential to provide design templates for the development of manufactured materials with similar properties, for applications such as biomedical implants and microfluidic “lab on a chip” devices.7
In order to better understand the chemical and topographic properties of these natural surfaces, a range of techniques has been applied in their analysis, including atomic force microscopy (AFM), scanning electron microscopy (SEM) and gas chromatography-mass spectrometry (GC-MS).4,5,6,8 The superhydrophobicity, and hence self-cleaning properties, of these surfaces has been shown to arise due to a combination of their surface chemistry and surface topography. In particular, the presence of hierarchical topography, comprising features on the nano- and micro-scale, contributes to their ability to repel water droplets. This, in turn, allows a pristine surface to be maintained by the droplets removing foreign particles as they roll across the surface. A common method for assessing the hydrophobicity of a surface is through the measurement of the “water contact angle”, which is the angle measured through the water at the three-phase (liquid/substrate/air) point of contact. A high contact angle indicates a low level of adhesion with water, and surfaces with a water contact angle over 150° are considered to be “superhydrophobic”. Certain plant leaves and insect wing membranes satisfy this criterion. Upon examination, many similarities have been shown to exist between the chemical and nano-structural properties of hydrophobic plant and insect cuticles, however, the outer surfaces of plant cuticles have been found to include a larger range of compounds, including aldehydes, ketones and fatty acids, whereas insect cuticle surfaces are predominantly comprised of long chain alkanes.
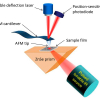
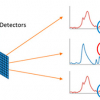
We have applied infrared microspectroscopy and imaging to the analysis of the spatially resolved chemical composition of the wing membrane of several insect species, including damselflies Ischnura heterosticta and Xanthagrion erythroneurum, dragonflies Diplacodes bipunctata and Diplacodes melanopsis, and the Clanger cicada Psaltoda claripennis.9,10 The sensitivity of Fourier transform infrared (FT-IR) microspectroscopy to the varied functional groups making up the insect wing membrane made it an ideal technique for probing the chemical properties of these structures at the micron scale, while SEM was used to examine the nanoscale topography of these membranes. Infrared microanalysis was undertaken at two synchrotron beamlines—the Infrared Microspectroscopy (IRM) beamline at the Australian Synchrotron, which operates a conventional point mapping Fourier transform infrared (FT-IR) microscope, and the IRENI IR imaging beamline at the Synchrotron Radiation Center (SRC), Madison, Wisconsin. By coupling an FT-IR microscope to a synchrotron source, significant advantages could be gained with regard to the intensity of the focused beam for single point FT-IR microanalysis of samples areas of the order of 3–5 µm2. Two-dimensional mapping of samples with this focused beam also allowed high spectral quality IR absorbance maps to be generated with diffraction limited spatial resolution, and this method was employed by the IRM beamline.11 The IRENI beamline at SRC, however, was designed to enable the use of a Focal Plane Array (FPA) IR imaging detector of 128 × 128 detector pixels in conjunction with a high magnification 74× microscope objective, which was made possible by the collection of twelve separate infrared beams from the SRC “ALADDIN” synchrotron storage ring.12 Although the SRC ALADDIN synchrotron ceased operation in 2014, the IRENI beamline enabled the high spatial resolution mapping of a range of materials and its success has led the planning of similar beamlines at other synchrotron facilities.

FT-IR spectroscopy and imaging of dragonfly and damselfly wing membranes
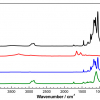
Dragonfly and damselfly wings were obtained from two sources. Fresh wings of the species D. bipunctata and I. heterosticta, were obtained from suburban regions of Melbourne, Australia, while aged samples of D. melanopsis and X. erythroneurum, collected in the 1970s, were provided by Museum Victoria, Australia. Scanning electron microscopy (FeSEM–Supra 40VP, Carl Zeiss GmbH, Jena, Germany) of gold-coated wing membranes showed that the surfaces of the Odonata wings possess an outer layer of nanometre-scale structures that are randomly distributed across the surface, as shown for the damselfly I. heterosticta in Figure 1A. While the overall pattern was found to be broadly similar between the four species, the size and separation of these structures was found to vary slightly between species. An FT-IR microanalysis of all four Odonata species wing membranes was conducted at the Australian Synchrotron IRM beamline, using a Bruker Hyperion 2000 IR microscope and V80v FT-IR spectrometer. Small sections of wing membrane, approximately 5 × 5 mm2, were mounted in a metal frame to support the edge of the sample and avoid movement of the delicate membrane during analysis. Spectra produced for all samples were consistent with the known composition of the insect cuticle (Figure 1B), with strong Amide I (1610–1695 cm–1) and Amide II (1480–1575 cm–1) absorption bands from the amide and amine groups of protein and chitin, as was a small C=O ester peak at 1735 cm–1, CH2 and CH3 stretching peaks (nCH2 and nCH3, 2840–3000 cm–1) from protein, chitin, alkanes and fatty acids, and broad overlying O–H and N–H stretching peaks obtained between 3200 cm–1 and 3400 cm–1. The most notable difference between the four species was the strength of the nCH peaks, in particular the symmetric and antisymmetric nCH2 peaks, with both damselfly species showing stronger nCH2 absorption than either of the dragonfly species. Two-dimensional spectral mapping of small areas of each species’ wings was performed using a 5 × 5 µm2 microscope aperture for 3 µm steps in both the x and y directions over a total area of 100 × 100 µm2. Figure 1C shows a plot of the integrated area of the antisymmetric nasCH2 peak between 2913 cm–1 and 2931 cm–1 across the wing membrane of the I. heterosticta damselfly, showing the heterogeneous nature of this distribution. Spectral subtraction of lower intensity nCH spectra from this 2D data set (black areas in Figure 1C) from higher nCH spectra (yellow areas in Figure 1C), confirmed that the main contribution to the higher nCH areas was from symmetric and antisymmetric nCH2 stretching (Figure 1D), indicating a heterogeneous distribution of long chain hydrocarbons such as waxes across the wing membrane. This is consistent with observations reported elsewhere, in which solvent extraction followed by the gas chromatography-mass spectrometry (GC-MS) analysis of wing membranes from the dragonfly Hemicordulia tau indicated the dominance of long chain alkanes of chain length from C10 to C34, with a smaller contribution (7%) being obtained from palmitic acid.8 Water contact angle measurements were made of wing membranes from each of the four species of dragonfly and damselfly studied, and the observed angles are shown against the IR absorbance spectra for each species in Figure 1B. These show a clear relationship between the long chain wax concentration, as indicated by the strength of the nCH2 peaks, and the water contact angle, showing that the species with higher CH2 absorbance show the greatest hydrophobicity, indicating a role for the chemical composition as well as the surface nanostructuring in promoting the superhydrophobic property of these insects’ wings.

FT-IR spectroscopy and imaging of cicada wing membranes
The native Australian cicada Psaltoda claripennis is shown in Figure 2A, examples of which were collected from Brisbane parkland areas. Sections of the wing membrane (approximately 5 µm in thickness) were then cut into squares approximately 5 × 5 mm2 and rinsed with Milli-Q H2O (resistivity of 18.2 MW cm–1) before being used for microanalysis.6 SEM of gold-coated wing membranes showed the presence of a highly regular pattern of “nanopillars” across the wing surface (Figure 2B), with an additional micron-scale texture also observed. It is these nanopillar structures that have been shown to be responsible for the bactericidal properties of these cicada wings, with the structures appearing to penetrate the membrane of bacteria that became attached to the surface. FT-IR spectroscopic imaging of the cicada wing membranes was conducted at the SRC IRENI beamline, Madison, Wisconsin.10 Wing samples were mounted into the same metal support frame used for the Odonata wings, and 64 × 64 pixel IR image frames were captured, each covering an area of 34.5 × 34.5 µm2. IR absorbance spectra of the cicada wings were found to be broadly similar to those obtained for the Odonata wings, showing the presence of spectral peaks attributable to protein, chitin, alkanes and fatty acids. Multiple image frames were tiled together to produce IR absorption images covering a larger area. Figure 3A–F shows IR absorption maps of two regions of wing, one covered by a 3 × 3 frame map (A, C and E) and one covered by a 4 × 1 frame map (B, D and F). In each case, an image of the Amide II distribution (integrated from 1483 cm–1 to 1588 cm–1) and nasCH2 distribution (2913–2931 cm–1) is shown. A third image is a Red:Green:Blue (RGB) composite in which the red and green channels are combined from the nasCH2 map, with the blue channel showing the Amide II. These images indicate that the Amide II peak shows a small amount of variation over the two image areas (Figure 3A and B) probably due to differences in wing membrane thickness, whereas the nasCH2 images (Figure 3C and D) show punctate distribution, with distinct features of a few microns in size seen as red spots in the nasCH2 map. These show up clearly as yellow spots in the combined RGB images (Figure 3E and F), indicating localised high concentrations of long chain hydrocarbons. Furthermore, a larger scale patterning or texture can be discerned over the areas of wing imaged, with CH2 “points” appearing to cluster into certain areas, while other areas show fewer spots. Figure 3G shows the CH stretch region (2800–3150 cm–1) of selected spectra from a high CH2 “spot” in Figure 3C (red trace) and a low CH2 area (black trace). The spectral subtraction of these traces is shown in Figure 3H, confirming that the high CH features in the IR images are due primarily to long chain aliphatic hydrocarbons, since only symmetric and antisymmetric nCH2 stretching peaks, along with a weaker C–H bending peak around 1470 cm–1 (not shown), were observed in the subtracted spectra.

In the case of the cicada wing, evidence could be seen of a hierarchical distribution of the hydrophobic wing surface coating. This combination of micron scale hydrocarbon features, arranged in patterns on the scale of tens of microns, may contribute to the high hydrophobicity of the cicada wing, which for P. claripennis has been shown to have a water contact angle of greater than 150° and is therefore considered to be superhydrophobic.
Conclusion
Insect wings possess some remarkable properties, enabling them to thrive in challenging environments. Often, a combination of nano- and micro-structured topography, with chemical heterogeneity on the micron scale, provides these structures with efficient superhydrophobic, self-cleaning and antibacterial capabilities. Their complex surfaces are providing inspiration for the design of novel coatings and surface structures that can mimic these beneficial properties. High spatial resolution FT-IR spectroscopy and imaging, when combined with other techniques such as SEM and GC-MS, provide unique insights into the complex chemical patterning that contributes to this functionality. Further developments in FT-IR imaging, such as 3D infrared tomography and scanning near-field nano spectroscopy13,14 have the potential to add a further dimension to the chemical and physical information that is enabling us to obtain a greater understanding of these natural surfaces. These techniques can also continue to be applied to the study of man-made material surfaces that attempt to emulate the properties of their biological templates.
References
- S.H. Nguyen, H.K. Webb, P.J. Mahon, R.J. Crawford and E.P. Ivanova, “Natural insect and plant micro-/nanostructured surfaces: an excellent selection of valuable templates with superhydrophobic and self-cleaning properties”, Molecules 19, 13614 (2014). doi: http://dx.doi.org/10.3390/molecules190913614
- W. Barthlott and C. Neinhuis, “Purity of the sacred lotus, or escape from contamination in biological surfaces”, Planta 202, 1 (1997). doi: http://dx.doi.org/10.1007/s004250050096
- T. Wagner, C. Neinhuis and W. Barthlott, “Wettability and contaminability of insect wings as a function of their surface sculptures”, Acta Zool. 77, 213 (1996). doi: http://dx.doi.org/10.1111/j.1463-6395.1996.tb01265.x
- J. Hasan, H.K. Webb, V.K. Truong, G.S. Watson, J.A. Watson, M.J. Tobin, G. Gervinskas, S. Juodkazis, J.Y. Wang, R.J. Crawford and E.P. Ivanova, “Spatial variations and temporal metastability of the self-cleaning and superhydrophobic properties of damselfly wings”, Langmuir 28, 17404 (2012). doi: http://dx.doi.org/10.1021/la303560w
- M. Sun, G.S. Watson, Y. Zheng, J.A. Watson and A. Liang, “Wetting properties on nanostructured surfaces of cicada wings”, J. Exp. Biol. 212, 3148 (2009). doi: http://dx.doi.org/10.1242/jeb.033373
- E.P. Ivanova, J. Hasan, H.K. Webb, V.K. Truong, G.S. Watson, J.A. Watson, V.A. Baulin, S. Pogodin, J.Y. Wang, M.J. Tobin, C. Löbbe and R.J. Crawford, “Natural bactericidal surfaces: Mechanical rupture of Pseudomonas aeruginosa cells by cicada wing”, Small 8, 2489 (2012). doi: http://dx.doi.org/10.1002/smll.201200528
- Y. Yoon, D. Kim and J.-B. Lee, “Hierarchical micro/nano structures for super-hydrophobic surfaces and super-lyophobic surface against liquid metal”, Micro Nano Syst. Lett. 2, 1 (2014). doi: http://dx.doi.org/10.1186/s40486-014-0003-x
- S.H. Nguyen, H.K. Webb, J. Hasan, M.J. Tobin, R.J. Crawford and E.P. Ivanova, “Dual role of outer epicuticular lipids in determining the wettability of dragonfly wings”, Colloid. Surface. B 106, 126 (2013). doi: http://dx.doi.org/10.1016/j.colsurfb.2013.01.042
- S.H. Nguyen, H.K. Webb, J. Hasan, M.J. Tobin, D.E. Mainwaring, P.J. Mahon, R. Marchant, R.J Crawford and E.P. Ivanova, “Wing wettability of Odonata species as a function of quantity of epicuticular waxes”, Vibr. Spectrosc. 75, 173 (2014). doi: http://dx.doi.org/10.1016/j.vibspec.2014.07.006
- M.J. Tobin, L. Puskar, J. Hasan, H.K. Webb, C.J. Hirschmugl, M.J. Nasse, G. Gervinskas, S. Juodkazis, G.S. Watson, J.A. Watson, R.J. Crawford and E.P. Ivanova, “High-spatial-resolution mapping of superhydrophobic cicada wing surface chemistry using infrared microspectroscopy and infrared imaging at two synchrotron beamlines”, J. Synchrotron Radiat. 20, 482 (2013). doi: http://dx.doi.org/10.1107/S0909049513004056
- P. Heraud, S. Caine, N.Campanale, T. Karnezis, D. McNaughton, B.R. Wood, M.J. Tobin and C.C.A. Bernard, Neuroimage 49, 1180 (2010). doi: http://dx.doi.org/10.1016/j.neuroimage.2009.09.053
- M.J. Nasse, M.J. Walsh, E.C. Mattson, R. Reininger, A. Kajdacsy-Balla, V. Macias, R. Bhargava and C.J. Hirschmugl, Nat. Methods 8, 413 (2011). doi: http://dx.doi.org/10.1038/nmeth.1585
- M.C. Martin, C. Dabat-Blondeau, M. Unger, J. Sedlmair, D.Y. Parkinson, H.A. Bechtel, B. Illman, J.M. Castro, M. Keiluweit, D. Buschke, B. Ogle, M.J. Nasse and C.J. Hirschmugl, “3D spectral imaging with synchrotron Fourier transform infrared spectro-microtomography”, Nat. Methods 10, 861 (2013). doi: http://dx.doi.org/10.1038/nmeth.2596
- H.A. Bechtel, E.A. Muller, R.L. Olmon, M.C. Martin and M.B. Raschke, “Ultrabroadband infrared nanospectroscopic imaging”, Proc. Natl. Acad. Sci. USA 111, 7191 (2014). doi: http://dx.doi.org/10.1073/pnas.1400502111