Laura Melton and Michael Taylor
Pesticides & Wildlife Science and Advice for Scottish Agriculture (SASA), Roddinglaw Road, Edinburgh, EH12 9FJ, UK. E-mail: [email protected]
This article is Crown copyright. All SASA images are subject to Crown copyright protection unless otherwise indicated. You may use and re-use Crown copyright images (not including logos) free of charge in any format or medium, under the terms of the Open Government Licence. Authorisation to reproduce any material identified as being the copyright of a third party must be obtained from the copyright holders concerned.
Poisoning investigations
In the United Kingdom, the Wildlife Incident Investigation Scheme (WIIS) investigates suspected poisoning of non-target vertebrate wildlife, livestock, companion animals and beneficial insects (essentially honey/bumble bees) when exposure to pesticides is suspected. In Scotland, the scheme is operated by Science and Advice for Scottish Agriculture (SASA), a scientific division of the Scottish Government.1 WIIS was set-up in the 1970s to monitor effects of persistent organochlorine insecticides on birds and has evolved since then to provide a means of post-registration surveillance of pesticide use as part of the pesticide registration process. The scheme also provides evidence for use by the Scottish Government and police to enforce the correct use of pesticides, identifying and penalising those who deliberately or recklessly misuse and abuse pesticides.
Poisoning incidents are usually categorised as follows:
- Approved use—where a pesticide is used in accordance with its conditions of authorisation but non-target species are still affected;
- Misuse—where the pesticide product has not been used according to the conditions of use, either carelessly or accidently, without the intention of harming non-target species;
- Abuse—where a pesticide (often a product that is no longer approved for use) has been used illegally in a deliberate attempt to poison animals;
- Unspecified use—where a pesticide is detected but the source of the chemical is unknown.
The number of suspected poisoning incidents investigated at SASA in 2011 was 237.2 Of these, only one incident resulted from the approved use of pesticides, three incidents resulted from misuse of pesticides and 16 incidents from abuse. In five incidents, pesticides were detected but it was not possible to establish the source of the chemical.
Pesticide analysis
The main challenges in pesticide residue analysis of samples associated with suspected poisoning incident investigations arise from:
- the wide range of specimens that need to be tested, such as animal liver and kidney tissues, digestive tract material, bait material, i.e. animal carcasses deliberately laced with pesticides and beneficial insects (for example, honeybees);
- the hundreds of pesticide products currently approved for use in the UK;
- products that are no longer approved for use in the UK but are still obtainable and their use has been abused by some individuals;
- the regular introduction of new products to the market;
- the need to comply with rigorous quality assurance procedures as the analytical results may be presented, scrutinised and challenged in enforcement and legal proceedings;
- analytical methods, techniques and instrumentation used must be capable of the detection, unequivocal identification and quantitation of the poison in a variety of complex matrices.
Until 2010, gas chromatography tandem mass spectrometry (GC-MS/MS) was used at SASA for multiple pesticide residue analysis in biological samples, but this was only useful for pesticides amenable to the technique. However, liquid chromatography tandem mass spectrometry (LC-MS/MS) was routinely used in the laboratory for the analysis of multiple pesticide residues that could be present in various fruit and vegetables. LC-MS/MS was also used for the analysis of biocides (i.e. anticoagulant rodentides) and other chemicals such as alphachloralose.3 So, it seemed long overdue for us to develop an LC-MS/MS method to complement the existing GC-MS/MS method and thereby significantly increase the WIIS pesticide target lists.

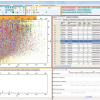
A new LC-MS/MS target list of 102 pesticides and metabolites was devised using Scottish pesticide usage data and records of chemicals implicated in UK poisoning incidents. The GC-MS/MS method that was already in place at SASA was based upon an ethyl acetate extraction with a gel permeation chromatographic (GPC) clean-up prior to analysis by GC-MS/MS. For the new method, a portion of the crude ethyl acetate extract was solvent exchanged into methanol and simply filtered prior to direct analysis by LC-MS/MS. Electrospray ionisation was achieved using a Quattro Ultima tandem (triple quadrupole) mass spectrometer. The instrument was operated in positive-ion mode. HPLC separation was achieved using gradient elution for screening. When a positive result was indicated following the initial [multiple reaction monitoring (MRM)] screen, a separate confirmation experiment was performed which included the original screen transition and an alternative MRM transition using a faster isocratic HPLC separation.
Chicken livers and chicken breasts were purchased from local retail outlets for use as pseudo-matrix material for recovery experiments and for preparation of matrix-matched calibration standards. A mixed pesticide standard solution was added to blank chicken liver and blank chicken muscle at 0.1 mg kg–1 and 1.0 mg kg–1 prior to the extraction stage in order to validate the method in accordance with European Union Analytical Quality control (AQC) guidelines for method validation and quality control procedures for the determination of pesticide residues in food and feed4 and in food of animal origin.5

Poisoning incidents
The new LC-MS/MS method is successfully and routinely used at SASA and, since its introduction in 2011, we have confirmed and refuted the presence of different pesticides in several non-target species that were believed to be victims of accidental or deliberate poisoning. This latter circumstance demonstrates the major benefit of using LC-MS/MS for multiple pesticide residue analysis, i.e. the rapid discovery of illegal practices. Our results, to date, indicate that the substance most abused in Scotland is the carbamate insecticide carbofuran, even though products containing this active ingredient lost their “approval for use” in the UK in 2001. Residues of carbofuran and its metabolite 3-hydroxycarbofuran have been detected in numerous poisoning incidents investigated by us during 2011, 2012 and 2013. Victims of carbofuran poisoning throughout this period include the common buzzard (Buteo buteo), golden eagle (Aquila chrysaetos), red kite (Milvus milvus), raven (Corvus corax), sparrowhawk (Acipitor nisus), domestic cats and pet dogs. Other pesticides and metabolites detected by LC-MS/MS include other carbamate insecticides aldicarb, aldicarb sulfoxide, bendiocarb and methiocarb.
Aldicarb was approved for use in the UK as an insecticide. It lost its approval status in 2007 and it is illegal for anyone to still have the substance in their possession. Approval for agricultural uses of bendiocarb were withdrawn in 2003. Pesticide formulations containing bendiocarb are, however, still approved for professional use for public health/hygiene purposes (for example, destruction of wasps’ nests). There are many amateur use products also available. Illegal use of more than one chemical together has frequently been seen in WIIS.
Bees
The increased target list also includes neonicotinoid insecticides (namely acetamiprid, clothianidin, imidacloprid, thiacloprid and thiamethoxam). Neonicotinoid insecticides have come under scrutiny recently due to concerns about their contribution to the decline in bee populations. In April 2013, the European Commission adopted a proposal to restrict the use of clothianidin, imidacloprid and thiamethoxam for a period of two years. The restrictions will apply from December 2013.

Reassuringly, at the time of publication of this article, no residues of neonicotinoids have been detected in bee samples submitted to SASA since 2011. In November 2012, however, the method was used to successfully detect and quantify a very low background residue of propiconazole (a fungicide used on many crops) in honeybees, which had not caused the death of the bees.
Conclusion
Since adopting the LC-MS/MS method in 2011, it continues to prove useful in providing quick, quantitative results in many different species.6,7
References
- http://www.sasa.gov.uk/wildlife-environment/wildlife-incident-investiga…
- http://www.sasa.gov.uk/document-library/pesticide-poisoning-animals-2011
- K. Hunter, M.J. Taylor, E.A. Sharp, L.M. Melton and S. LeBouhellec, “Determination of chloralose residues in animal tissues by liquid chromatography-electrospray ionisation tandem mass spectrometry”, J. Chromatogr. B 805, 303 (2004). doi: 10.1016/j.jchromb.2004.03.015
- Method Validation and Quality Control Procedures for Pesticide Residues in Food and Feed, Document SANCO/12495/2011, ec.europa.eu/food/plant/plant_protection_products/guidance_documents/docs/qualcontrol_en.pdf
- European Union CD 2002/657/EC
- www.sasa.gov.uk/document-library/positive-results-2012
- M.J. Taylor, L.M. Melton, E. Sharp and J.E. Watson, “A liquid chromatography-electrospray tandem mass spectrometry method for the determination of multiple pesticide residues involved in suspected poisoning of non-target vertebrate wildlife, livestock and pets”, Anal. Methods 5(1), 248 (2013). doi: 10.1039/C2AY25555C