Mario Thevis and Wilhelm Schänzer
Center for Preventive Doping Research—Institute of Biochemistry, German Sport University Cologne, Am Sportpark Müngersdorf 6, 50933 Cologne, Germany. E-mail: [email protected] and [email protected]
Introduction
Doping control authorities and sports drug testing laboratories are frequently confronted with the illicit use of performance-enhancing therapeutics and therefore various analytical strategies have been developed to detect a misused drug and/or its metabolic product(s) in blood or urine specimens. Besides the administration of clinically approved drugs prohibited in sports, new drug candidates currently undergoing early or advanced clinical trials have also been the subject of investigations concerning their prevalence and abuse by athletes.1 One class of emerging therapeutics includes the so-called selective androgen receptor modulators (SARMs), which is a chemically diverse group of non-steroidal biologically active compounds that tissue-selectively activate the androgen receptor. These substances, being developed as medications for cancer cachexia and sarcopenia as well as other uses including, e.g., male contraception,2 have demonstrated considerable potential to enhance performance and increase muscle size and strength in proof-of-concept studies. Although none of these drug candidates has yet received clinical approval or has been launched by pharmaceutical companies, doping control laboratories are urged to screen for these compounds as recent findings have provided evidence for their easy availability.3
Arylpropionamide-derived SARMs
The clinically most advanced SARMs comprise an arylpropionamide-based nucleus with varying substituents at the A- and B-rings (see Figure 1), which were first reported in 1998 by Dalton and co-workers.4 Andarine (also referred to as S-4, Figure 1, structure 1) has demonstrated great therapeutic potential and underwent phase-II clinical trials as well as extensive in vitro and in vivo metabolism studies. These showed significant deacetylation and hydroxylation as well as other degradation reactions followed by sulfonation and glucuronidation. Metabolically more stable derivatives were considered bearing cyano and halogen substituents instead of nitro and N-acetyl residues (as present in the case of Andarine) yielding analogues such as S-1, S-22 or S-24 (Figure 1, structures 2, 3 and 4, respectively), and medically beneficial results triggered an increased attention and desire of certain “athletes”, who discussed availability, dosing, desirable as well as presumably undesirable effects for several years in Internet chat rooms. In 2005, concerns about the misuse of illegally produced and/or obtained SARMs by professional and amateur athletes grew among anti-doping authorities, initiating doping control research programmes supported and pursued by international anti-doping organisation and research groups. These yielded a detection method for SARMs bearing the above mentioned arylpropionamide nucleus using liquid chromatography-tandem mass spectrometry.5 In the following years, various additional SARMs with core structures derived from quinoline, tetrahydroquinoline or bicyclic hydantoin nuclei were studied in detail and comprehensive mass spectrometric data were generated enabling the detection and identification of the intact analytes in urine or blood specimens.6

Metabolism of arylpropionamide-derived SARMs
Measuring active drugs is an excellent approach if blood (as well as plasma or serum) samples are tested. However, since urine represents the most commonly collected doping control matrix, concise knowledge of the extent of metabolism of these therapeutics is required to ensure the detection of SARMs after use, especially in a retrospective context concerning anti-doping analysis. Consequently, in vitro metabolism studies using human liver microsomal preparations were conducted with Andarine and other arylpropionamide-based SARMs.7 These revealed a variety of potential target compounds for sports drug testing purposes, especially the B-ring depleted Andarine (Figure 1, structure 5). This further complemented existing doping control methods and broadened the spotlights of drug testing laboratories for this new class of non-steroidal anabolic agents.

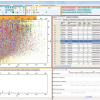
![Figure 3. ESI product ion mass spectra of deprotonated molecules [M – H]– of Andarine (a) and the contaminant (b) in a black market product.](/sites/default/files/articles/MS-22_4-F3.jpg)
First findings of SARMs on the black market
Despite the fact that SARMs are still undergoing clinical trials and are not yet approved in any country, a growing concern regarding illegally produced and/or distributed SARMs has led to the inclusion of the entire class of these emerging drugs into the prohibited list as issued by the World Anti-Doping Agency in 2008. The necessity of this action was corroborated by recent findings of Andarine being advertised and sold via the Internet (Figure 2).3 Numerous Internet-based companies have tried to attract customers to buy their products with “SARM-like properties”, and authentic Andarine was indeed detected in one of these cases. The product termed S-4 in accordance with scientific publications was labelled to contain 300 mg S-4 per mL of ingestion solution. Qualitative analyses revealed the presence of Andarine as proved by liquid chromatography-high resolution/high accuracy (tandem) mass spectrometry (Figure 3a). However, the amount of Andarine was quantified as 150 mg mL–1 only and, most importantly, an unknown impurity accounting for approximately 15% was observed. Based on in-depth elucidation of the dissociation pathway of Andarine after negative electrospray ionisation (ESI) followed by collision-induced dissociation, the origin of the product ions (Figure 3a) was established, and fragments were assigned to distinct parts of the molecule. As such, the ions at m/z 150, 205, 261 and 289 represent the intact B-ring, A-ring and chiral core structure of Andarine, respectively, as shown in Figure 3a. Using these data in combination with accurate masses of precursor and product ions of the contaminant as well as information on general synthetic routes towards arylpropionamide-based SARMs, the contamination was identified as 2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-phenyl)-3-(4-nitro-3-trifluoromethylphenylamino)-propionamide (S-4–2A) resulting from incomplete purification of a synthesis intermediate and, consequently, duplication of the A-ring in the final compound (Figure 3b). The intact molecule of S-4–2A was found at m/z 495, 55 Da higher than Andarine. In addition, the ion at m/z 150 was missing, which indicated a modification of the B-ring, corroborated by the fact all product ions representing the A-ring and core structure (i.e., m/z 205, 261 and 289) were observed. High resolution mass spectrometry at a resolving power of 100,000 did not allow the separation of two species contributing to the signal at m/z 205 (which would account for m/z 150 + 55) and the accurate mass measurement of m/z 495 further supported the expected elemental composition of S-4–2A with C18H13N4O6F6 (error = 1.8 ppm). The effect on the human organism of this contamination is not known and is considered a major health risk if administered!
Metabolism of Andarine after oral administration
As a consequence of the obviously easy availability of black market products of SARMs, urine samples containing the active drug Andarine and its metabolites were received in the doping control laboratory of Cologne in 2010. Although not representing a controlled administration study, the specimens were used to study and compare the metabolites’ profile to those products predicted by means of earlier in vitro incubations.8 The most abundant metabolites of Andarine are summarised in Figure 4, matching those observed and characterised using human liver microsomal preparations. Major metabolic pathways included hydroxylation, deacetylation, reduction and conjugation to glucuronides or sulfates, giving rise to a great variety of degradation products. The elimination of the entire B-ring resulting in the metabolite M5 yielded a target analyte of great utility due to its comparably long excretion period and, thus, option to detect the administration of Andarine several days after it had ceased to be used. M5 was further generated in amounts sufficient for nuclear magnetic resonance spectroscopy characterisation using electrochemical metabolism simulation systems that predominantly produced the B-ring depleted analogue to Andarine.9 After confirmation of the predicted composition and structure, M5 (or its glucuronic acid conjugate M4) was chosen as the main analyte for routine doping control analyses to indicate the illicit administration of Andarine.

Conclusions
Selective androgen receptor modulators represent a novel class of therapeutics with promising clinical properties, particularly concerning their use for the treatment and prevention of many illnesses, including cancer cachexia, sarcopenia, debilitating diseases and osteoporosis. Compared to conventional steroid replacement therapies for which undesirable effects are observed particularly due to the lack of tissue selectivity, the use of SARMs has proved efficacy and safety in proof-of-concept studies and advanced clinical trials. These properties, however, have also resulted in a great potential for their misuse in both amateur and elite sport. Shortly after first research data on SARMs were published in scientific articles, numerous discussions in Internet-based chat rooms on “how to use” and “where to obtain” could to be followed, indicating the considerable interest of persons other than patients in these new therapeutic agents. Consequently, sports drug testing authorities and laboratories have begun implementing new target compounds in routine screening assays to detect illicit applications before these SARMs are officially launched and introduced into the pharmaceutical market. These preventive approaches shall minimise the “window of opportunity” that cheating athletes might have when obtaining and administering non-approved and hardly available experimental drug candidates to illegally improve their performance.
References
- M. Thevis, A. Thomas, M. Kohler, S. Beuck and W. Schänzer, J. Mass Spectrom. 44, 442 (2009).
- M.L. Mohler, C.E. Bohl, A. Jones, C.C. Coss, R. Narayanan, Y. He, D.J. Hwang, J.T. Dalton and D.D. Miller, J. Med. Chem. 52, 3597 (2009).
- M. Thevis, H. Geyer, M. Kamber and W. Schänzer, Drug Test. Analysis 1, 387 (2009).
- J.T. Dalton, A. Mukherjee, Z. Zhu, L. Kirkovsky and D.D. Miller, Biochem. Biophys. Res. Commun. 244, 1 (1998).
- M. Thevis, M. Kamber and W. Schänzer, Rapid. Commun. Mass Spectrom. 20, 870 (2006).
- M. Thevis and W. Schänzer, J. Mass Spectrom. 43, 865 (2008).
- T. Kuuranne, A. Leinonen, W. Schänzer, M. Kamber, R. Kostiainen and M. Thevis, Drug Metab. Dispos. 36, 571 (2008).
- M. Thevis, A. Thomas, G. Fußhöller, S. Beuck, H. Geyer and W. Schänzer, Rapid Commun. Mass Spectrom. 24, 2245 (2010).
- M. Thevis, W. Lohmann, Y. Schrader, M. Kohler, W. Bornatsch, U. Karst and W. Schänzer, Eur. J. Mass Spectrom. 14, 163 (2008).