Frank Vanhaecke and Luc Moens
Department of Analytical Chemistry, Ghent University, Krijgslaan 281–S12, BE-9000 Ghent, Belgium. E-mail: [email protected]
Introduction
Inductively coupled plasma-mass spectrometry (ICP-MS) was introduced commercially in 1983 as a very sensitive analytical technique to be deployed for (ultra)trace element analysis. Compared to the previously existing techniques of atomic absorption spectrometry (AAS) and ICP-optical emission spectrometry (ICP-OES), the main advantages offered by ICP-MS over these techniques were its pronounced multi-element capabilities and substantially higher detection power, respectively. As a mass spectrometric device, ICP-MS, however, also provides isotopic information on the analyte elements and this feature can be taken advantage of for further extending the application range of ICP-MS and for tackling real-life problems for which elemental information does not provide sufficient information. While the “standard” ICP-MS equipment—equipped with a quadrupole filter or a sector field mass spectrometer for mass analysis and a single ion detector only—can be deployed for studying induced and relatively large natural variations in the isotopic composition of target elements, multi-collector (MC) MC-ICP-MS—equipped with a sector field mass spectrometer and an array of ion detectors, allowing simultaneous detection of the intensity of the ion beams corresponding to all isotopes of a target element—is a dedicated tool for highly precise isotopic analysis, also permitting one to trace down extremely small natural variations. Real-life applications carried out at the Department of Analytical Chemistry of Ghent University and based on the use of both single- and multi-collector instrumentation will be used in this article to illustrate the wide application range of isotopic analysis via ICP-MS.
Measuring induced variation
As a result of a thorough mixing of all nuclides before the formation of the earth, it can be roughly stated that all elements display an isotopic composition that can be considered as “constant” (more accurately, only varies within very narrow limits) in all terrestrial materials. This statement will be refined further below. As a result of this, anthropogenically induced changes (e.g., the enrichment of mined uranium to produce material containing a large enough fraction of 235U for use as fuel in a fission reactor or for the production of nuclear weapons) can be easily identified, while on the other hand, material with an anthropogenically changed isotopic composition can be used as a standard in an elemental assay using isotope dilution or as a tracer suited for studying (bio)chemical reactions or physical processes.
During the enrichment of U, mentioned above, depleted uranium (DU, depleted in 235U compared to mined uranium) is produced as a waste product. As a result of its very high density (almost twice that of lead), DU is used for the production of ammunition, capable of penetrating the armoured steel of military vehicles and for counter-weights mounted in the tail and the wings of airplanes to improve their stability. The fact that upon impact, uranium rapidly inflames, is considered an advantage in the former case, self-evidently not in the latter. On the basis of measurement of the 235U/238U isotope ratio, ICP-MS permits natural, depleted or enriched U to be discerned from one another (while in the latter case, also the enrichment grade can be determined). In 1992, an El Al freight plane crashed into an apartment block in Amsterdam (The Netherlands). Since rescue workers that had been active on the site of the disaster complained about persistent health problems, rumours of the effect the inhalation of DU had on their health were raised. However, isotopic analysis of the U present in their urine gave no indication whatsoever of the presence of DU at the time of sampling.1
The ability to measure isotope ratios also allows the use of isotope dilution (ID) as a calibration strategy in ICP-MS, alongside of the more common approaches of external calibration and the method of standard additions. In this approach, a known amount of spike—a standard solution of the analyte element itself, but displaying an isotopic composition substantially different from the natural one—is added to a known amount of sample, and an isotope ratio of the target element is measured in the sample, spike and mixture solutions. Based on these data, the target element concentration in the sample can be calculated. An isotope ratio is far more robust than a signal intensity, because it is much less affected by changes in the instrumental operation conditions or changes in the matrix composition; this makes ID a very reliable approach. When carried out with the utmost care and control of all factors affecting the final uncertainty, ID-MS provides highly accurate and precise results and therefore, institutions producing reference materials are very keen users of this calibration strategy. Also in routine applications, its use may be very suitable, especially if the sample pre-treatment includes a trace/matrix separation step. This is because once isotopic equilibration is established (when the nuclides originating from the sample and from the spike are mixed to such an extent that it is impossible to identify where a given nuclide is originally stemming from), analyte loss does not affect the final result. As a result, incomplete or even variable recovery of the target element during the separation process is not a threat to the accuracy of the results, as long of course as a sufficient quantity of element is retained for a reliable isotope ratio measurement. This strategy was, for example, used for the determination of ultra-trace amounts of Fe in highly concentrated AgNO3 solutions, intended to be used in the production of photographic emulsions.2 After addition of a 54Fe spike, the bulk element Ag was precipitated by adding highly pure HBr. After filtering off the AgBr precipitate, the Fe concentration in the filtrate could be determined based on Fe isotope ratio measurement. Although co-precipitation of Fe with the huge amount of AgBr formed was not unlikely, this did not affect the final result.
Tracer experiments with stable isotopes rely also on the fact that elements occurring in nature display the “standard” isotopic composition, such that the addition of a tracer with a markedly different isotopic composition followed by isotope ratio measurements can be used to study a physical process or a chemical reaction. When studying the transport of an element across a membrane, for example, element determination suffices if this transport is unidirectional (Figure 1). If the transport, however, is bidirectional, then element determination does not provide a full picture, whereas the use of an isotopic tracer and isotopic analysis of the target element at both sides of the membrane does. This is exactly the approach that was used for studying the transport of Mg—an essential element for all living beings—over the epithelial tissue of tilapia fish (Oreochromis mossambicus, used as a model species).3 Epithelial tissue was mounted in a holder and was in contact at both sides with a solution containing 0.5 mmol L–1 of Mg (Figure 2). At one side, the composition of the solution was such that it mimicked lumen (the content of the intestine), on the other side, the composition was such that it mimicked blood. The potential difference over the membrane was continuously monitored and nullified in order to avoid this from acting as a driving force. Samples were taken at regular time intervals from both sides and the isotopic composition of the Mg determined. From the results thus obtained, biologists could study the transport process and calculate its rate.
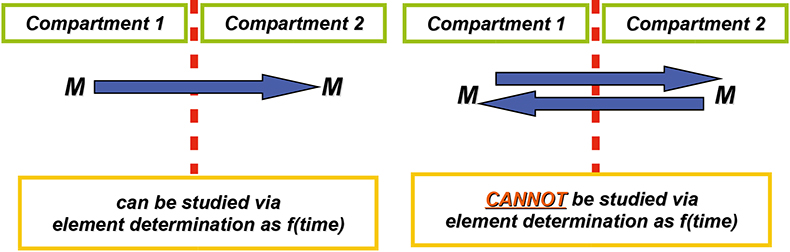
Figure 1. Unidirectional transport of an element across a membrane (left) can be studied via element determination; bidirectional transport (right) cannot be studied in this way and requires isotopic analysis to be documented completely.
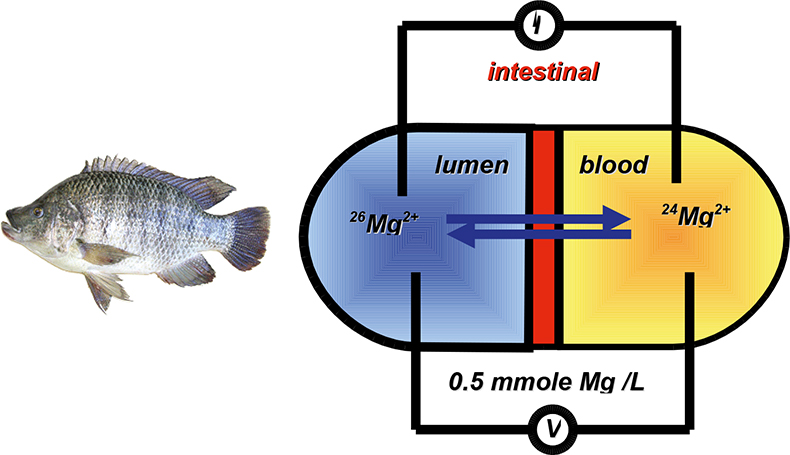
Figure 2. Experimental set-up for studying the bidirectional transport of Mg2+ across the intestinal epithelium of tilapia fish (Oreochromis mossambicus). The potential across the membrane is continuously monitored and nullified. Samples are taken from both compartments at regular time intervals for determination of the isotopic composition of Mg.
A similar approach was also used for studying the uptake of metals by Daphnia magna. This is a small aquatic crustacean, often studied in ecotoxicology, because its well-being is related to the environmental conditions, while the animals are easy to culture in the lab and show a constant response to toxic substances, such as metals. Daphnids can ingest these metals via two pathways—via the respiratory system (the gills) and via the alimentary channel. For a considerable time, it was assumed that only metals present in algae (their food) and taken up via the alimentary channel affected the health of these animals, but more recently, exposure studies demonstrated that also waterborne Zn has an adverse effect on the reproduction of the daphnids.4 The relative importance of both exposure routes can be determined via the use of isotopic tracers, whereby the Zn present in the algae is enriched in one isotope of Zn and that present in the water is enriched in another one (Figure 3). Isotopic analysis of exposed animals for their isotopic composition of Zn reveals the relative uptake via both routes.5
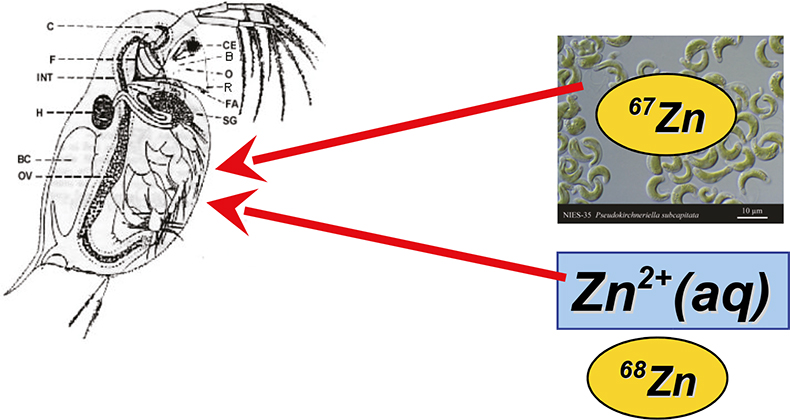
Figure 3. Evaluation of the relative importance of the two potential exposure routes (via the respiratory system and via the alimentary channel) by means of a tracer experiment using stable isotopic tracers. Zn dissolved in the water is enriched in one Zn isotope, that incorporated in the algae in another.
Measuring natural variation
One reason by which elements show a deviation from “constant isotopic composition” is the presence of radiogenic isotope(s), the isotopic abundance(s) of which increases/increase very slowly as a result of the decay of a naturally occurring long-lived radionuclide(s). This can be illustrated using Sr as an example. Sr has four isotopes—84Sr, 86Sr, 87Sr and 88Sr—one of which (87Sr) is enriched by the radioactive decay of 87Rb, which is converted into 87Sr as a result of the emission of a beta-particle. The half-life of 87Rb is almost 50 billion years (approximately 10 times longer than the age of the earth). While the isotopic composition of Rb changes over geological times, it is at present at the same level for all terrestrial materials. The isotopic composition of Sr, on the other hand, does show variation and is determined by the time that Sr “has spent in the presence” of Rb and the relative concentrations of the two elements. Pb is another well-known example, as no less than three of its four isotopes are the end-product of radioactive decay of naturally occurring nuclides of U or Th. As a result of the factor “time”, the combination of element determination and isotopic analysis can be used for dating purposes (determination of the age of geological materials), as already deployed by geologists for decades, using thermal ionisation mass spectrometry (TIMS) for isotope ratio measurements.
As for Sr, it is accepted that the element is transferred without measurable fractionation from the bedrock to the soil, from the soil to the vegetation and from the vegetation to the animals grazing on this vegetation, Sr isotopic analysis can also be used for provenancing of agricultural products (e.g., wine, cheese). By comparing the isotopic composition of Sr (or the 87Sr/86Sr isotope ratio) in such a product with that of the potential geographical regions of origin (showing different geologies), information on the provenance of the product can be obtained and forgeries (e.g., wine not coming from the region indicated on the bottle) can be tracked down. Also the origin of humans can be studied in this way. Human tooth tissue consists of two main materials: an outer shiny part, enamel, is formed during childhood and is the hardest substance in the human body, and an inner, much softer part, dentine, which is continuously renewed (Figure 4). The isotopic composition of the Sr in the enamel of an individual adult is thus governed by that of the Sr present in the food consumed during his/her childhood, while that of the Sr present in the dentine is governed by the isotopic composition of the Sr in the food consumed during the past few years. Especially for studying archaeological human relicts, this difference is important, as in modern times people consume foodstuffs originating from various parts of the world. This approach was, for example, used at Ghent University for studying gravefield populations, excavated at a historically important basilica in The Netherlands. Isotopic analysis of Pb can be utilised also for provenance determination purposes, as Pb from different mining areas may display considerably different isotopic fingerprints, and this approach has been used in a wide variety of contexts, ranging from environmental studies, with as a goal deciphering which fraction of atmospheric Pb derives from petrol-Pb, to archaeological studies, aimed at obtaining a more profound insight into the origin of the raw material used for the manufacturing of objects of art.6
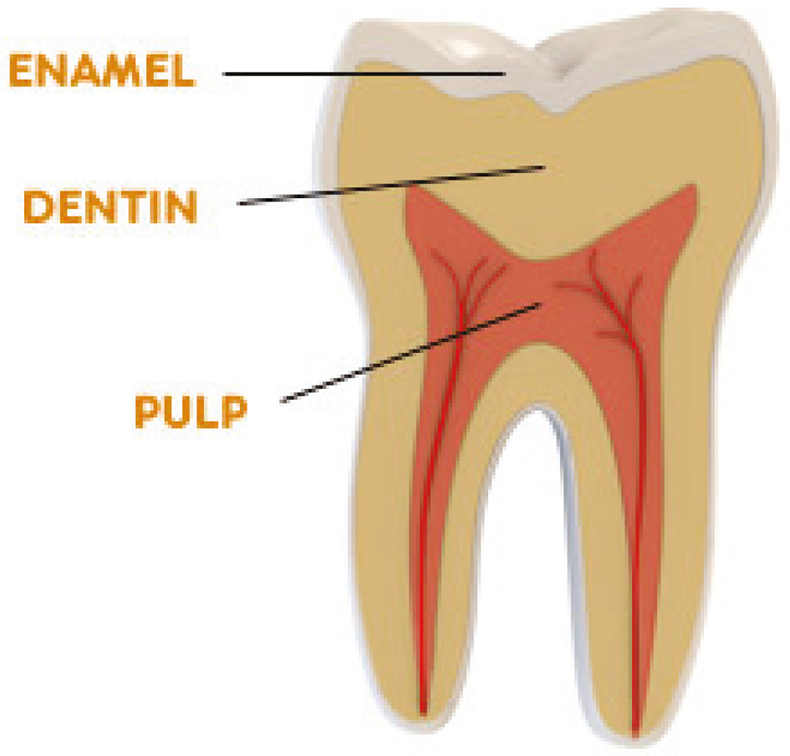
Figure 4. Diagram of a tooth (molar) showing the enamel and dentine fractions.
Other elements show natural variation in their isotopic composition as a result of the occurrence of isotopic fractionation, meaning that the isotopes of an element do not take part with the same efficiency in physical processes or chemical reactions, as a result of differences between them in equilibrium state (thermodynamics) or reaction rates (kinetics). This effect is most pronounced for lighter elements that abundantly take part in all kind of processes, such as H, C, N, O and S. This is widely exploited for all kinds of applications, based on the use of gas source mass spectrometry. In particular, with the introduction of multi-collector ICP-MS, such fractionation effects—though of much smaller magnitude—are being revealed for an increasing fraction of the elements, including metals such as Fe, Zn or Sn. The combination of the ease with which these elements are ionised into M+ ions in the high-temperature ICP (ionisation temperature ~7500 K) and the high precision offered by the simultaneous determination of all ion beams involved (Figure 5) has led to these discoveries. While some practical applications are being developed already, such as the diagnosis of the severity of the occurrence of the blood disease hemochromatosis7 or the investigation of the origin of environmental contamination,8 it is clear that a more thorough understanding of these fractionation effects still needs to be gathered and practical applications based on their existence need to be developed.

Figure 5. Multi-collector array of a multi-collector ICP-MS instrument, allowing the simultaneous monitoring of the ion beams of all isotopes of a target element. The individual detectors can be moved to their correct position (motorised) and by means of electrostatic lenses, the beams can be steered into the detector entrances (courtesy of Thermo Fisher).
It must be also pointed out that obtaining accurate results with MC-ICP-MS is less self-evident than it may appear from the text above. The isotope ratio as measured needs to be corrected for what is called instrumental mass discrimination, referring to the fact that there is a difference in the efficiency with which isotopes of the same element are transferred from the ICP to the ion detector. As the concomitant matrix affects the extent of mass discrimination, the target element needs to be isolated from the matrix when aiming at detecting and quantifying small differences in isotope ratios, for example, of the order of 0.01% only. As a result of the dramatic effect that contamination may have on an isotope ratio, this separation process is preferably or necessarily (depending on the target element and the variation expected) carried out in a high quality clean laboratory, having a vastly lower number of atmospheric particles per unit of volume than in ambient air.
It is clear that the fact that ICP-MS—in contrast to AAS and ICP-OES—readily provides isotopic information is undeniably a very important advantage, providing benefits both in everyday applications and opening new areas of application. Hence this field of research is growing rapidly.
References
- J.A. Bijlsma, P. Slottje, A.C. Huizink, J.W.R. Twisk, G.B. van der Voet, F.A. de Wolff, F. Vanhaecke, L. Moens and T. Smid, Nephrol. Dial. Transplant. 23, 249 (2008). https://doi.org/10.1093/ndt/gfm461
- L. Balcaen, I. Geuens, L. Moens and F. Vanhaecke, Anal. Bioanal. Chem. 377, 1020 (2003). https://doi.org/10.1007/s00216-003-2177-6
- G. De Wannemacker, A. Ronderos, L. Moens, F. Vanhaecke, M. Bijvelds and Z. Kolar, J. Anal. At. Spectrom. 16, 581 (2001). https://doi.org/10.1039/b101183i
- K. De Schamphelaere, M. Canli, V. Van Lierde, I. Forrez, F. Vanhaecke and C.R. Janssen, Aquat. Toxicol. 70, 233 (2004). https://doi.org/10.1016/j.aquatox.2004.09.008
- L.I.L. Balcaen, K.A.C. De Schamphelaere, C.R. Janssen, L. Moens and F. Vanhaecke, Anal. Bioanal. Chem. 390, 555 (2008). https://doi.org/10.1007/s00216-007-1620-5
- F. Vanhaecke, L. Balcaen and P. Taylor, in ICP Spectrometry and its Application, 2nd Edition, Ed. by S.J. Hill. Sheffield Academic Press, Sheffield, p. 160 (2007).
- P.A. Krayenbuehl, T. Walczyk, R. Schoenberg, F. von Blanckenburg and G. Schulthess, Blood 105, 3812 (2005). https://doi.org/10.1182/blood-2004-07-2807
- C. Cloquet, J. Carignan, M.F. Lehmann and F. Vanhaecke, Anal. Bioanal. Chem. 390, 451 (2008). https://doi.org/10.1007/s00216-007-1635-y











