Introduction
Glow Discharge Mass Spectrometry (GDMS) is one of the most powerful solid state analytical methods for the direct determination of traces, impurities and depth profiling of solids.1–5 Glow discharge mass spectrometers, which are commercially available with fast and sensitive electrical ion detection, allow direct trace elemental determination in solid materials with good sensitivity and precision in the concentration range lower than ng g–1.6
The glow discharge is a low energy plasma (Figure 1) created by inserting two electrodes in a cell filled with gas at a low pressure (0.1–1 Torr). For analytical applications, argon is most commonly used. When a potential difference of the order of 1 kV is established between the two electrodes, “gas breakdown” happens, i.e. the gas is split up into positive ions and electrons resulting in the formation of plasma. The Ar+ ions formed in the glow discharge are accelerated toward the sample cathode and the sample material is sputtered at the cathode surface by ion bombardment. Sputtered atoms and molecules are ionised in the glow discharge plasma (negative glow) by Penning and/or electron impact ionisation and charge exchanges processes. A mathematical model of processes in a direct current glow discharge has been developed by Bogaerts and Gijbels.7 The positively-charged ions formed in the argon plasma of the glow discharge source are extracted and accelerated into the mass spectrometer, where the ion beams are separated according to their mass-to-charge and energy-to-charge ratio. The separated ions are electrically detected by a photomultiplier or channeltron detector.
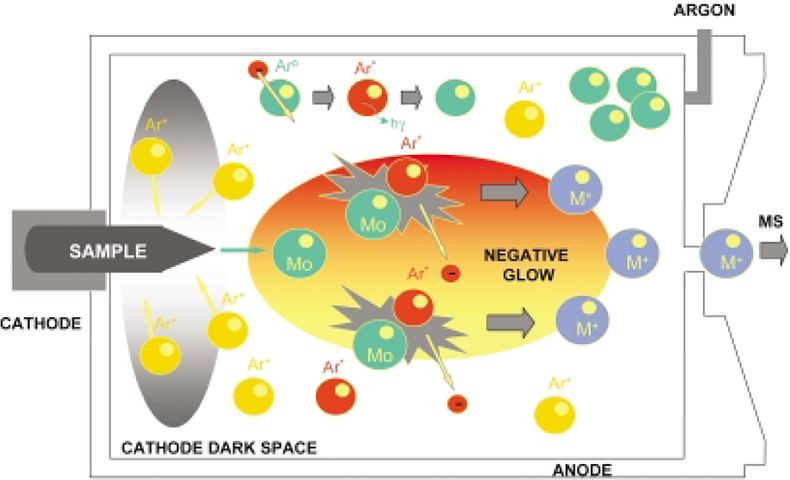
Figure 1. Schematic picture of the main processes occurring in a glow discharge.
The glow discharge ion source has been interfaced to most of the standard mass spectrometer types. The first commercially-available GDMS instrument used a double focusing magnetic sector mass analysis system, permitting the acquisition of high-resolution spectra with high sensitivity.8 Quadrupole mass spectrometers are typically more compact and less expensive than magnetic sectors. As a consequence, quadrupoles have been employed for fundamental and development research of GDMS,9,10 finally resulting in the commercial availability of a quadrupole GDMS system.11 Promising results have also been obtained from the coupling of glow discharge ion trap mass spectrometric sytems,12 double and triple quadrupole instruments,13 time-of-flight mass spectrometers14 and Fourier transform mass spectrometers.15,16 However, the commercially-available GD mass spectrometers presently only employ double-focusing and quadrupole-based mass spectrometers.
GDMS has found extensive application for multi-elemental determination in high-purity metallic and semiconducting bulk samples.17–19 Direct solid mass spectrometry techniques have major advantages with respect to sample preparation: few sample preparation steps are required, and the risk of contamination is significantly lower. Quantification of the analytical signal in solid mass spectrometry may be difficult, especially if no suitable standard reference material with the same matrix is available. However, GDMS is relatively free from matrix effects due to the separation of atomisation and ionisation phenomena in time and space during the sputtering of the sample. Screening data can be obtained by GDMS even when reference materials are not available. A simple comparison of the element signal intensity of the analyte with the element sensitivity of a reference element, defined as the ratio between the signal intensity and the elemental concentration, results in an accuracy of about 30%.20
One of the main challenges for glow discharge spectroscopy to overcome is the intrinsic requirement of the sample to be electrically conducting. Nevertheless, non-conducting solid samples can be directly analysed by using a radio frequency powered source,21 with direct current device plasma using the secondary cathode technique22,23 or by mixing the sample with a conducting host.24,25 When a secondary cathode with a circular orifice in its centre is employed, it is placed on top of the analytical specimen. Upon initiation of the discharge, the sputtering process is naturally concentrated in the inner edge of the conductive electrode, and a metallic layer is eventually produced on the sample surface. This electrically-conductive layer promotes sputtering of the sample, and the continuous deposition of the metallic layer allows extended plasma operation. This approach is limited by the geometry of the sample. It tends to be complex in terms of the optimisation of the discharge conditions in order to balance sputtering of the sample and of the secondary cathode. The application of a conducting binder that is mixed with the non-conducting powdered sample before the pressing of the homogenous mixture to an electrode is very common. The physical nature of the sample can affect the efficiency with which the mixture can be ground to a size fine enough to ensure a stable sample disk and plasma. Both techniques for the analysis of non-conductors by dc GDMS may present additional disadvantages. The formation of new disturbing molecular ions (argides, oxides from the secondary cathode or binder material) in mass spectra can be observed. The detection limits increase due to the dilution of the powdered sample with the conductor binder or due to the secondary cathode material (contributing to the blank). The preparation of the mixed electrode requires a grinding step, which could cause contamination. Other problems arise from trapping water vapour and atmospheric gases in the sample during the compaction process. In order to avoid these difficulties, radio frequency powered GD sources were introduced for the direct analysis of non-conductors.26–28 Despite extensive research performed in this field,1,2,26–32 it must be pointed out that no commercial source exists for a complete rf-GDMS system, even if rf devices have been sampled by a very wide range of mass analyser types. Among these, single quadrupole,28,33 ion trap,33 Fourier transform ion cyclotron resonance (FT-ICR),34 time-of-flight35 and double focusing instruments36,37 are worthy of mention.
As a surface analytical technique, GDMS can be used for the determination of element concentration as a function of sputtered depth. However, depth profile of thin layers using GDMS has had a subordinated role compared with GD-OES (GD-Optical Emission Spectrometry). In fact, to date most depth profile work has been carried out with optical emission instruments. The major reason for this is probably the fact that commercial GDMS devices are slow and cumbersome to operate compared with GD-OES instruments.38 However, a substantial amount of depth profile analysis has also been carried out using GDMS. With the development of a faster GDMS such as the one recently developed by Dorka et al., GDMS should be used more frequently for depth profiling39 in the future.
In the field of nuclear research and technology the chemical characterisation of different types of nuclear fuels, cladding materials, nuclear-waste glasses and smuggled nuclear samples, from the point of view of trace, major and minor elements as well as their isotopic composition, is of great importance. These materials can be analysed using several techniques based on nuclear and non-nuclear methods that can be, to a varying degree, tedious and time consuming. In the last year, non-nuclear methods based on MS have become predominant for the characterisation of samples of nuclear concern. The application of ICP-MS has been widely investigated40–43 for fission products and actinide determination as well as thermal ionisation MS (TIMS) for the routine analysis of isotopes in liquid samples.44 GDMS has also been used for the chemical characterisation of nuclear samples.45–48 In these applications, GDMS provides information on the chemical composition much faster than other techniques, making it possible to modify fuel production procedures and reactor conditions or to quickly recognise smuggled materials. The advantages of GDMS are low limits of detection, uniform element sensitivity and capability to measure all elements and even isotopes.
Instrumentation
For handling nuclear materials, difficulties arising from the radioactive nature of samples have to be overcome. The operator has to be protected from the radioactive material, which means that the use of glove-boxes is necessary. In addition, in order to avoid contamination of the working area, the analytical instrument has to be modified so that containment is assured and no radioactive material leaks either into the laboratory or into the environment. Complete instruments cannot be introduced into a glove-box because electronics are very sensitive to radiation, only the sampling stage takes place inside. Only a GD mass spectrometer (VG9000, Thermo Elemental) has been integrated in a glove-box for the characterisation of nuclear material.45 The glove-box encloses the ion source chamber, the interlock and the associated pumped system. In Figure 2, a schematic diagram of the installation of the GD source housing in the frame of the glove-box is given. The installation of the VG 9000 GDMS into the glove-box is described in detail by Betti et al.45
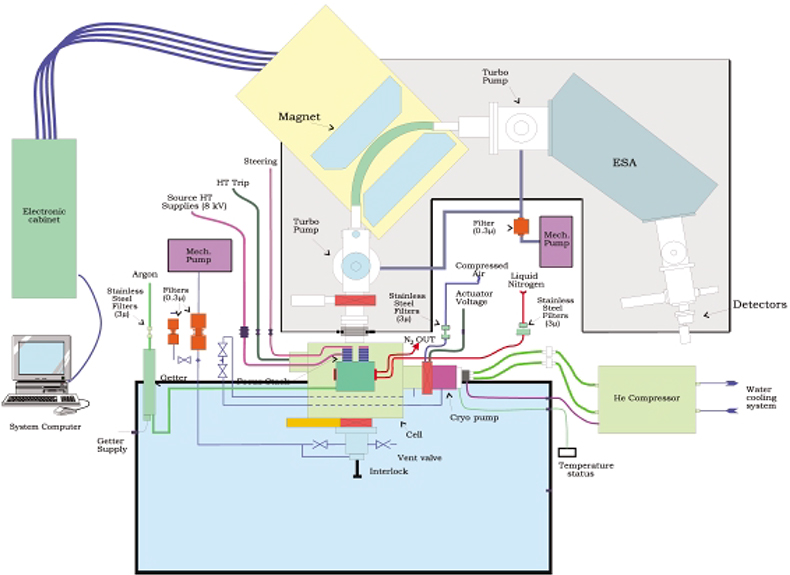
Figure 2. Schematic diagram of the installation of a GDMS in a glove box.60
Applications for nuclear samples
In our laboratory, a great deal of experience has been acquired in the use of GDMS for the chemical and isotopic characterisation of samples of nuclear concern. Plutonium and uranium oxide specimens, mixed uranium and plutonium oxide (MOX) and metallic fuels, simulated high burn-up nuclear fuels (simfuel), Zircaloy cladding materials, nuclear-waste glasses and smuggled nuclear materials have been investigated using GDMS. Some examples of conducting and non-conducting samples of nuclear concern characterised by a dc GDMS are discussed below.
Non-conducting nuclear samples
Two different approaches have been used for the analysis of non-conducting samples. For flat samples, a secondary cathode is placed directly in the front of the sample. The second approach, for powdered samples, consists in mixing a pure conductive host matrix, namely pure silver with tantalum or titanium in a ratio of 1 : 3. In Table 1, a list of the oxide-based nuclear fuels analysed as for trace elements is given. The percentage of oxygen ranges from about 12 to 18% m/m. Oxygen, as a major matrix element, causes severe problems due to its release from the oxide during discharge processes. Once released into the GD plasma, it influences the analytical signal by quenching excitation and ionisation agents.49 Moreover, in GDMS problems also arise from the presence of polyatomic oxides that create spectral interferences and give lower analytical sensitivity. “Getter metals” such as titanium or tantalum bond strongly with oxygen and reduce its availability to form oxides with the analytes or to quench metastable argon atoms.
Table 1. Non-conducting oxide-based samples. Composition given in % m/m for each component.48
Sample | Oxygen | Uranium | Plutonium | Cerium | Neptunium |
U3O8 | 15.2 | 84.8 | — | — | — |
UO2 | 11.8 | 88.2 | — | — | — |
PuO2 | 11.8 | — | 88.2 | — | — |
(U,Pu)O2 | 11.8 | 49.3 | 38.9 | — | — |
(U,Pu)O2 | 14.9 | 61.8 | 23.3 | — | — |
(U,Pu)O2 | 13.8 | 73.8 | 12.4 | — | — |
(U,Pu)O2 | 13.2 | 82.3 | 4.5 | — | — |
(Pu,Ce)O2 | 17.9 | — | 35 | 45 | — |
(U,Pu,Np)O2 | 13.8 | 46.1 | 35 | — | 4.1 |
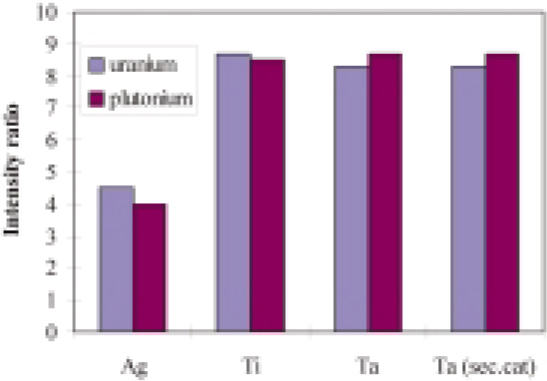
Figure 3. U+ :UO+ and Pu+ : PuO+ intensity ratios from oxide-based nuclear samples obtained with different binder materials and a tantalum secondary cathode; m/z values measured were 238, 254, 239, 255.48
In Figure 3, the ratios U+ : UO+ and Pu+ : PuO+ obtained with different binder materials are shown. Titanium and tantalum bind the oxygen stronger than silver, fewer UO+ and PuO+ ions are formed in the case of silver. The Relative Sensitivity Factors, RSFs, for the three binders for some selected trace elements of nuclear interest such as boron, lithium, cadmium and gallium are presented in Table 2. RSFs depend on the host matrix and in the case of silver higher RFS are obtained. This can be explained by the fact that the oxygen in the plasma is not gettered by the silver and also by the fact that a large quantity of the uranium and plutonium in the plasma are oxide species. This reduces the number of metal ions in the plasma available for ionisation.
Table 2. RSF values for uranium and plutonium dioxide using different metals as host matrices and secondary cathode.48
| Host matrix | Secondary cathode | ||||||
Ag | Ti | Ta | Ta | |||||
UO2 | PuO2 | UO2 | PuO2 | UO2 | PuO2 | UO2 | PuO2 | |
11B | 1.25 ± 0.30 | 1.33 ± 0.25 | 0.98 ± 0.20 | 1.04 ± 0.30 | 0.97 ± 0.20 | 1.05 ± 0.20 | 0.98 ± 0.30 | 0.98 ± 0.28 |
7Li | 2.01 ± 0.23 | 1.98 ± 0.30 | 1.12 ± 0.13 | 1.08 ± 0.20 | 1.15 ± 0.28 | 1.12 ± 0.25 | 1.13 ± 0.20 | 1.10 ± 0.20 |
114Cd | 1.97 ± 0.30 | 2.00 ± 0.28 | 0.87 ± 0.20 | 0.90 ± 0.25 | 0.90 ± 0.28 | 0.88 ± 0.25 | 0.90 ± 0.25 | 0.93 ± 0.28 |
69Ga | 1.58 ± 0.15 | 1.63 ± 0.13 | 0.95 ± 0.10 | 0.98 ± 0.12 | 0.95 ± 0.12 | 0.95 ± 0.13 | 0.98 ± 0.10 | 1.02 ± 0.10 |
For a secondary cathode, tantalum was chosen, and it was found that its property as a getter for oxygen is also an advantage. Indeed, it was found that the U+ : UO+ and Pu+ : PuO+ ratios obtained with a tantalum secondary cathode are of the same order of magnitude as those obtained with tantalum and titanium binders (Figure 3). The main result of the investigation is that for several elements, the RSFs were found to depend on the oxygen content in the sample (Figure 4). Therefore, a specific matrix reference sample for the quantitative analysis of oxygen containing samples is necessary.48
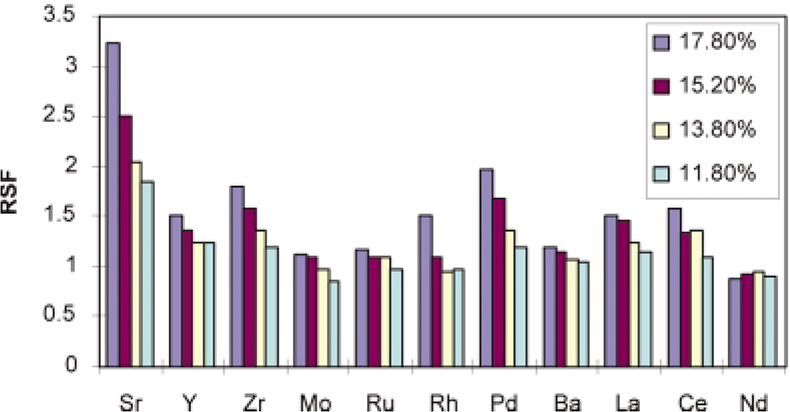
Figure 4. RSF values of some trace elements in uranium and plutonium dioxide samples as function of oxygen content. Measurements performed using a tantalum secondary cathode.48
Materials for nuclear reactor fuel preparation need to be characterised for both the isotopic composition of the major elements and the concentration of trace elements. The acceptable levels of impurities in fresh nuclear fuels vary according to the characteristics of the reactor. In order to monitor contaminations during the fabrication process, the determination of the trace elements should be performed in the starting material as well as in the final pellets of fresh fuels. For these measurements, analytical methods with proven reliability, accuracy and precision are necessary. Among currently available techniques, dc GDMS and quadrupole ICP-MS have been used successfully.50 In Table 3, the results obtained analysing a uranium oxide reference sample (Morille, CEA, France) using standard and matched-matrix RSFs are reported. To obtain results with the highest possible accuracy, matrix-specified RSFs values are required. The data presented in Table 3 demonstrate the high stability of the discharge when using the secondary cathode technique for the analysis of uranium oxide samples. Typical precisions in the order of 10% RSD or better can be obtained. For elements with concentrations of 5–10 µg g–1, an analytical precision higher than 10% RSD was obtained. Using matrix specific RSFs, an accuracy of 5% was generally obtained.
In GDMS, detection limits, in the absence of any interference, are calculated from signals equalling three times the noise of the background signal. For uranium and plutonium oxides analysed with the secondary cathode technique, detection limits for several trace elements are at the low µg g–1 level, as shown in Table 3, when using an integration time of 120 ms per isotope. As can be seen from this table, the detection limits for Fe, Cu, Ni, Mo, Ti, W and Zr are higher because the blank contribution of these elements stemming from the tantalum mask has to be taken into account.
Table 3. GDMS quantitative analysis of Morille uranium oxide reference sample based on matrix-specific and standard RSF.50
Element | Certified value (µg g–1) | Matrix-specific RSF (µg g–1) | Bias (%) | RSD (%) | Standard RSF (µg g–1) | Bias (%) | Detection limit (µg g–1) |
Ag | 10.4 ± 1.6 | 10.2 ± 1.3 | 1.9 | 12.1 | 9.3 ± 2.3 | 10.8 | 0.1 |
Al | 99 ± 6 | 87 ± 5 | 12.1 | 5.5 | 87 ± 3 | 11.9 | 0.5 |
Ba | 3.8 ± 1.6 | 3.5 ± 1.5 | 7.9 | 40.8 | 2.5 ± 0.8 | 35.5 | 0.2 |
Ba | 9.6 ± 0.4 | b |
|
| 11.4 ± 0.3 | –18.8 | 0.7 |
Bea | 5.4 ± 0.6 | 3.8 ± 0.4 | 29.6 | 10 | b |
| 0.5 |
Bi | 24.4 ± 1.9 | 20.9 ± 1.7 | 14.3 | 7.7 | 41 ± 3 | –68 | 0.6 |
Caa | 93 ± 8 | 94 ± 9 | –1.1 | 9.1 | 95.8 ± 4.2 |
| 0.4 |
Cd | 4.9 ± 0.7 | 5 ± 0.4 | –2 | 7.6 | 3.4 ± 1 | 30.6 | 0.5 |
Co | 9.8 ± 2 | 11.1 ± 0.8 | –13.3 | 6.9 | 9.5 ± 0.3 |
| 1.3 |
Cr | 99 ± 2 | 102 ± 5 | –3 | 4.7 | 94 ± 11 | 4.7 | 1.9 |
Cu | 50.2 ± 1 | 52.1 ± 3.3 | –3.8 | 6 | 63 ± 7 | –25.6 | 0.6 |
Dy | 0.5 ± 0.06 | c |
|
|
|
| 0.7 |
Eu | 0.52 ± 0.03 | c |
|
|
|
| 0.5 |
Fe | 211.6 ± 6.5 | 207.2 ± 10.8 | 2.1 | 5 | 313 ± 22 | –47.9 | 2.4 |
Gd | 0.56 ± 0.06 | c |
|
|
|
| 0.9 |
In | 9.4 ± 1 | 10.4 ± 0.5 | –10.6 | 4.6 | 8.1 ± 0.3 | 14.3 | 1.0 |
Mg | 19.3 ± 1.5 | 19.4 ± 1.6 | –0.5 | 7.9 | 12.2 ± 1 | 36.8 | 0.1 |
Mn | 24.5 ± 0.5 | 29.3 ± 1.1 | –19.6 | 3.6 | 30 ± 1 | –22.4 | 1.4 |
Mo | 147 ± 5 | 144 ± 9 | 2 | 6 | 175 ± 11 | –19 | 0.9 |
Ni | 147 ± 3 | 142 ± 4 | 3.4 | 2.7 | 143 ± 25 | 2.7 | 6.2 |
Pb | 101 ± 3 | 103 ± 9 | –2 | 8.3 | 111 ± 7 | –9.9 | 0.4 |
Sia | 100 ± 8 | 93 ± 6 |
| 6.1 | 245 ± 11 | –145 | 0.1 |
Sm | 0.5 ± 0.12 | c |
|
|
|
| 0.9 |
Sn | 18.5 ± 5.6 | 20.8 ± 3 | –12.4 | 13.7 | 15.3 ± 4.6 | 17.3 | 0.4 |
Th | 6.2 ± 0.8 | b |
|
|
|
| 0.4 |
Ti | 49.2 ± 2.6 | 48.6 ± 8 | 1.2 | 15.7 | b |
| 1.4 |
V | 48.7 ± 2.8 | 47 ± 2 | 3.5 | 4.1 | 50 ± 1 | –2.6 | 0.7 |
W | 100 ± 9 | 106 ± 11 | –6 | 9.9 | 95 ± 3 | 4.8 | 2.1 |
Zn | 98.6 ± 5.5 | 102 ± 10 | –3.4 | 9.3 | 148 ± 8 | –50 | 0.8 |
Zr | 59.9 ± 4.1 | 64 ± 7 | –6.8 | 10.4 | b |
| 0.9 |
*Bias (%) = (certified value – GDMS value) × 100 / certified value. a: Possible interferences: 9Be: 36Ar4+; 10,11B: 40Ar4+H, 40Ar4+; 40,41,42,43,44Ca: 40Ar+, 40ArH+, 12C14N16O+, 12C16O2+; 28,29,30Si: 56Fe2+, 27AlH+, 14N2+, 12C14N, 12C16OH+, 14N16O+; b: not determined; c: below detection limit
Plutonium oxide standards are not commercially available and one approach for the determination of bias is to apply different analytical techniques. The analysis of uranium and plutonium dioxides, using tantalum as secondary cathode, has shown that the RSF values are very similar in both matrices.48 This fact indicates that accurate GDMS results can be obtained using RSF values from a matrix of similar composition. The results for the Pu samples were obtained by using RSF values derived for a uranium oxide matrix. This confirms that both U oxide and Pu oxide matrices have the same behaviour in a glow discharge source. The quantitative analysis by ICP-MS was performed by using a multi-standard addition in order to obtain the most accurate and precise results possible. In Table 4, the results obtained by GDMS and ICP-MS in the analysis of a plutonium oxide specimen are shown. As can be seen, good agreement exists between the results obtained by both techniques.50
Table 4. Mean concentration (mg kg–1 PuO2) and intervals of confidence, IC, (%) referring to the 95 % confidence level (n = 25).50
Element/isotope | GDMS | ICP-MS | ||
| mg kg–1 | IC (%) | mg kg–1 | IC (%) |
Na | 1.32 | 22.0 | 0.99 | 36.2 |
Mg | 0.70 | 68.6 | 1.06 | 42.4 |
Al | 3.88 | 13.4 | 3.73 | 6.5 |
Fe | 0.91 | 73.6 | 1.75 | 54.3 |
234U | 19.80 | 1.0 | 19.40 | 44.4 |
235U | 638.00 | 1.1 | 637.00 | 1.2 |
236U | 170.00 | 1.8 | 171.00 | 4.9 |
237Np | 122.00 | 1.6 | 111.00 | 7.6 |
Isotopic composition for nuclear samples
In nuclear technology and generally in nuclear research, the precise and accurate measurement of isotope ratios is of great interest. The widely accepted method for their determination is TIMS. In this method, the sample must be dissolved and chemical separation of the analyte of interest is required before the analysis. GDMS has also been exploited for the determination of the isotopic composition in samples of nuclear concern.51 By comparison with TIMS, GDMS has turned out to be a competitive technique for the determination of the isotopic abundance of B and Li, as well as U, in terms of precision and accuracy. GDMS has the advantage of reduced handling of the sample before the analysis. For the determination of the isotopic composition of Pu, corrections for 238U and 241Am are necessary.
Radioisotopes traces in environmental samples
The movement of radionuclides in the environment has been studied for many years, with the principle objective of tracing the routes by which they accumulate in the food chain and become available for human consumption. The explosions in Reactor No. 4 of the nuclear power station of Chernobyl provide a point source for distribution of radionuclides and a unique opportunity to trace the mechanisms by which they are distributed. GDMS has been exploited for the determination of traces of uranium in soil samples.52 Caesium, strontium, plutonium, uranium and thorium in soils, sediments and vegetation have also been investigated.53 More recently, dc GDMS has been exploited for the determination of 237Np in Irish Sea sediments.54
The method applied for the analysis of non-conducting samples, using a secondary cathode, has also been employed for environmental samples. According to this method, the samples (soils, vegetation and sediments) need to be pressed into disc shaped electrodes. Discs obtained without blending with silver powder were found to be too fragile. Therefore, 5% of silver powder was added to the samples. This concentration was the minimum amount necessary to obtain stable disc electrodes without too much dilution. Stable discharge was obtained using a tantalum secondary cathode during the analysis. The combination of the use of blending material and a secondary cathode has been used for all environmental types of samples mentioned above.
As for the determination of elements and radioisotopes, at the trace level the sensitivity is an important parameter. The procedures based on GDMS are mainly affected by interferences arising from the sample matrix, the blending material and the discharge gas. Interferences from the matrix and discharge gas can be eliminated by using a high mass resolution. This normally results in a decrease of sensitivity, and hence is not suitable for trace element determinations. The blending material (the so-called conductive host matrix) can be chosen according to the specific sample requirements, or the latter can be neglected.
For example, in the analysis of traces of Th, the addition of silver can be a necessity. Silver produces a spectral interference due to the formation of 107Ag109Ag16O+, and a mass resolution of at least 1000 is necessary to separate the peak of this species from the Th peak. With the uranium isotopes, silver does not produce any interferences. Tantalum, however, gives rise to the formation of 180Ta40Ar16O+ and 181Ta40Ar16O+. These two polyatomic ions interfere with 235U+ and 236U+. When working at a mass resolution of 300, 180Ta40Ar16O+ can be resolved from those of 235U+ as well as 181Ta40Ar16O+ from 236U+. For the separation of signal from 180Ta40Ar16O+ and 236U+, respectively, a mass resolution of 1700 is necessary. The determination of 236U is of great importance because this isotope indicates the presence of irradiated uranium in the sample. For 236U, detection limits in the pg g–1 range or even less often are requested. They can be obtained by the use of a secondary cathode consisting of gold. In the case of 237Np determination, the most important interference is due to the formation of 181Ta40Ar16O+ at mass 236.9069, and a resolution of at least 1700 is necessary to separate this from the 237Np peak at mass 237.0482. In Figure 5, the GD mass spectrum for 237Np and 181Ta40Ar16O+ in a NIST Peruvian 237Np doped soil at a working mass resolution of 5500 is shown.
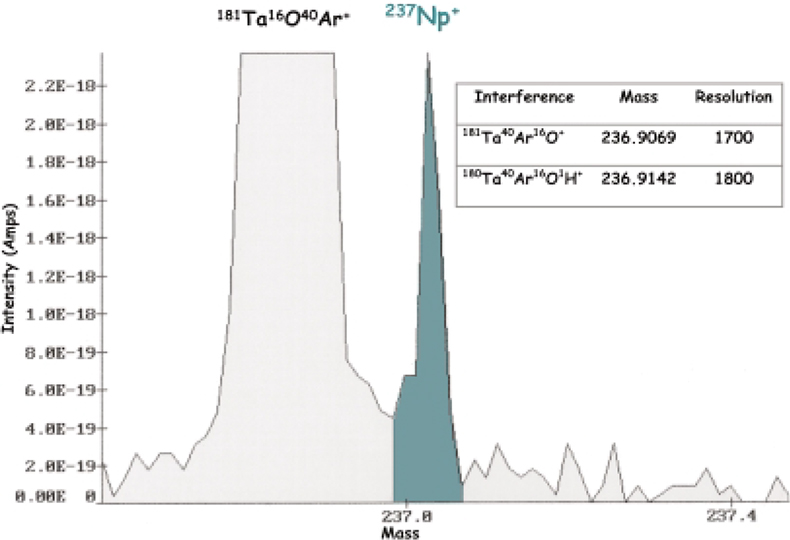
Figure 5. GD mass spectrum for 237Np in a NIST Peruvian doped soil. 237Np concentration is 2 ng g–1 at a mass resolution of 5500. Integration time is 4.5 s.53
Depth profiling of ZrO2 layers deposited on Zircaloy nuclear fuel cladding material
36% of the electrical energy produced in Europe comes from nuclear power plants.55 The major issues are the competitiveness of nuclear energy compared with other energy sources, the safety of nuclear reactors and the management of nuclear waste. The actual trend is to increase the burn-up of the nuclear fuel for economic reasons and to reduce the production of waste. One of the major effects encountered is the corrosion of the Zircaloy cladding used for containment of the fuel pellets.
In pressurised water reactors (PWR), the lithium hydroxide added to the primary coolant as alkaliser, in concentrations varying between 2 and 4 µg g–1 depending on the number of the reactor cycles,56 may increase the oxidation rate of the Zircaloy.57 In addition, boric acid is introduced at a concentration level of 1000 µg g–1 of boron in the cooling water of western PWR to control the core reactivity at the beginning of each reactor cycle. It has been found that this additive has a retarding effect on the formation of the ZrO2 corrosion layer.58 In order to evaluate the influence of different additives on the corrosion induced by the lithium it is necessary to study their incorporation in the layer of ZrO2 formed during reactor operation.
GDMS has also been used for the study of the mechanisms of corrosion of Zircaloy cladding of nuclear fuels, measuring the diffusion of the impurities in ZrO2 layers by depth profiling.59 It has been found that the use of a secondary cathode is essential. As shown in Figure 6(a), representing a depth profile for zirconium oxide layer deposited on aluminium, an anomalous peak of zirconium appears centred at about 600s in the metal/oxide interface. This behaviour was found to be due to instability of the plasma arising from the change in electrical conductivity of the system when passing from the insulating nature of the oxide layer to the conductive oxide/ metal interface. This problem was overcome by applying a secondary cathode. In Figure 6(b), three well-defined zones are shown to be present: the oxide layer (0–1100 s), the metal oxide interface (1100–1700 s) and the metallic support (1700–3000 s). The profile for lithium was also registered. Figure 6(b) shows that a fairly constant concentration of Li is found in the oxide layer, then it decreases in the interface zone and goes down to zero in the metallic phase.
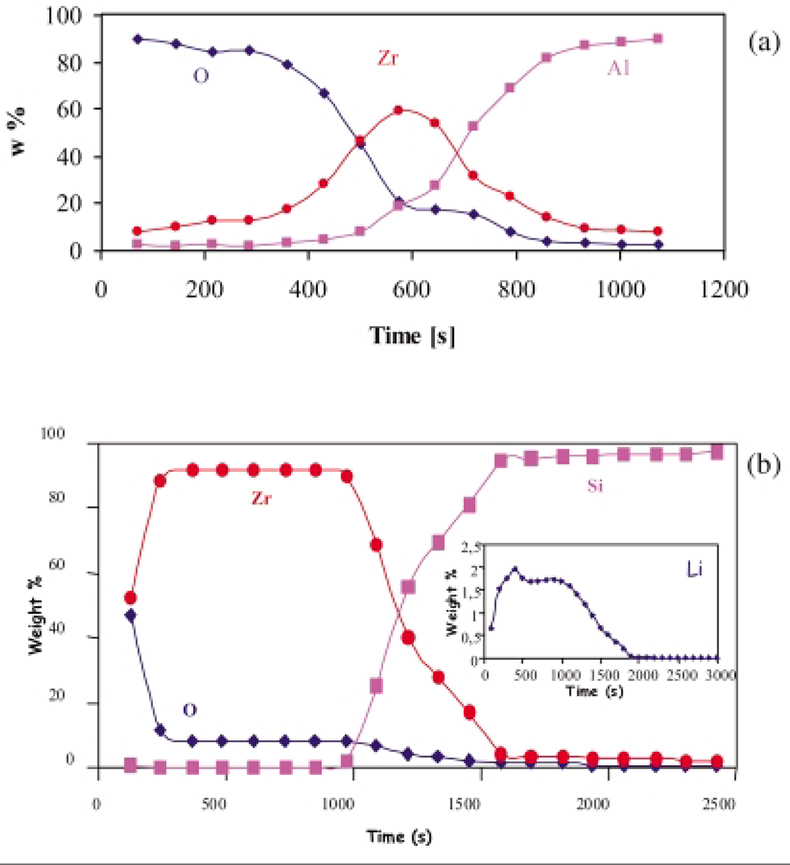
Figure 6. (a) GDMS depth profile of a thin layer of ZrO2 deposited on Al.59 (b) GDMS depth profile for the major elements and lithium of a thin layer of ZrO2 deposited on silicon and doped with lithium.59
By GDMS depth profiling, the effect of B on Li diffusion has been also studied. The influence of boron solution additions considerably reduces the oxidation of Zircaloy provoked by lithium.59 The large reduction of the sample weight gain has now been corroborated with the depth profiles obtained by GDMS. Figures 7(a) and 7(b) compare the profiles of lithium incorporated in the oxide layer after 7 days of reaction in autoclave with 1000 mg kg–1 of Li and 1000 mg kg–1 of Li + 1000 mg kg–1 of B, respectively. The depth profiles show a reduction of lithium uptake of one order of magnitude during corrosion in presence of boron. An interesting property noted for boron is that this element does not penetrate in the oxide layer, as is evident from Figure 7(b). This indicates an external protective effect, e.g. poisoning of adsorption sites or formation of a thin protective overlayer on the surface. The possible buffering effect of the boric acid controlling the pH of the solution seems less probable for the simple fact that any other pH buffer could also produce the same beneficial effect on the oxide growth.
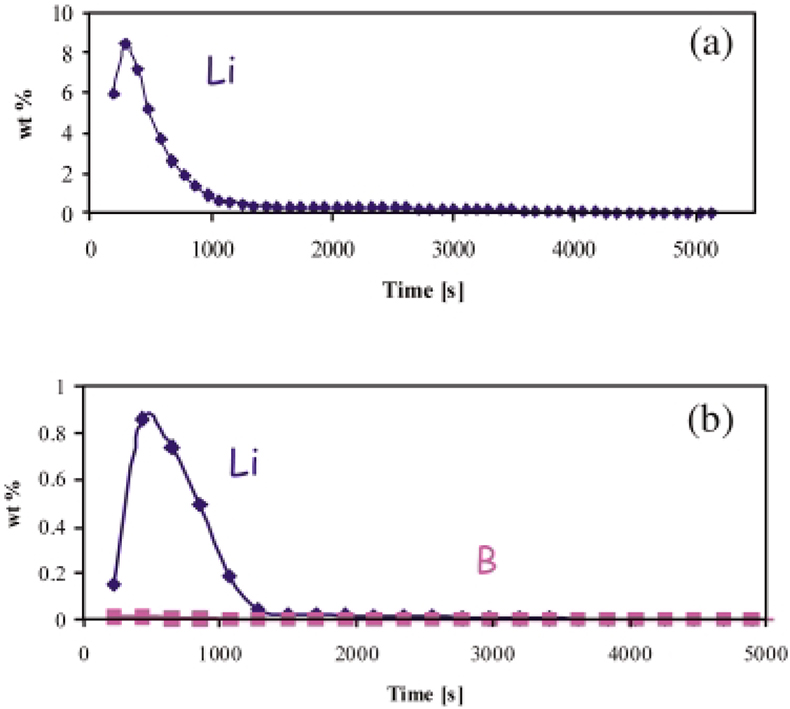
Figure 7. Lithium depth profile for a ZrO2 layer obtained in autoclave simulating nuclear reactor operating conditions, (a) without and (b)with addition to the corrosive media of boron (1000 ng g–1).59
Conclusions
GD-based techniques have been shown to be of great use for bulk and depth profiling measurements in the case of conductive as well as non-conductive samples, with an unrivalled flexibility of applications. In particular, GDMS has been successfully applied to the characterisation of samples of nuclear concern. Its “non-destructive” nature and the fast sample preparation make the technique very attractive for the characterisation of radioactive samples. Moreover, major, minor and trace elements can be determined in the same analysis. GDMS has been demonstrated to be competitive with TIMS for determination of the isotopic compositions of matrix elements. The method can also be used for the determination of traces of radioisotopes in environmental matrices, exploiting the technique of the secondary cathode. Mechanisms of the diffusion of trace elements can be followed by depth profiling analysis.
References
- R.K. Marcus and J.A.C. Broekaert (Eds), Glow discharge plasmas in analytical spectroscopy. John Wiley & Sons (2003).
- R.K. Marcus (Ed.), Glow Discharge Spectroscopies. Plenum, New York (1993). https://doi.org/10.1007/978-1-4899-2394-3
- R. Gijbels, M. Van Straaten and A. Bogaerts, Adv. Mass. Spectrom. 13, 241 (1993).
- W.W. Harrison, W. Hang, X. Yan, K. Ingeneri and C. Schilling, J. Anal. At. Spectrom. 12, 891 (1997). https://doi.org/10.1039/a701641g
- A. Bogaerts and R. Gijbels, Fresenius J. Anal. Chem. 364, 367 (1997). https://doi.org/10.1007/s002160051352
- J.S. Becker and H.J. Dietze, Spectrochim. Acta B 53, 1475 (1998). https://doi.org/10.1016/S0584-8547(98)00110-4
- A. Bogaerts and R. Gijbels, Spectrochim. Acta B 53, 1 (1998). https://doi.org/10.1016/S0584-8547(97)00122-5
- J.E. Cantle, E.F. Hall and C.J. Turner, Int. J. Mass Spectrom. Ion Processes 46, 11 (1983). https://doi.org/10.1016/0020-7381(83)80040-0
- C.G. Bruhn, B.L. Bentz and W.W. Harrison, Anal. Chem. 50, 373 (1978). https://doi.org/10.1021/ac50024a050
- N. Jakubowski, D. Sütwer and G. Tölg, Int. J. Mass Spectrom. Ion Processes 71, 183 (1986). https://doi.org/10.1016/0168-1176(86)85055-8
- R.C. Hutton and A. Raith, J. Anal. Atom. Spectrom.7, 623 (1992). https://doi.org/10.1039/ja9920700623
- S.A. McLuckey, D.E. Goeringer, K.G. Asano, G. Vaidyanathan and J.L. Stephenson Jr, Rapid. Commun. Mass Spectrom. 10, 287 (1996). https://doi.org/10.1002/(SICI)1097-0231(199602)10:3<287::AID-RCM429>3.0.CO;2-H
- F.L. King and W.W. Harrison, Int. J. Mass Spectrom. Ion Processes 89, 171 (1989). https://doi.org/10.1016/0168-1176(89)83059-9
- D.P. Myers, M.J. Heintz, P.P. Mahoney, G. Li and G.M. Hieftje, Appl. Spectrosc. 48, 1337 (1994). https://doi.org/10.1366/0003702944028029
- R.K. Marcus, P.R Cable, D.C. Duckworth, M.V. Buchanan, J.M. Pochkowski and R.R. Weller, Appl. Spectrosc. 46, 1327 (1992). https://doi.org/10.1366/0003702924123683
- C.H. Watson, C.M. Barshick, J. Wronka, F.H. Laukien and J.R. Eylier, Anal. Chem. 68, 573, (1996). https://doi.org/10.1021/ac9508412
- F.L. King and W.W. Harrison, Mass Spectrom. Rev. 9, 285 (1990). https://doi.org/10.1002/mas.1280090303
- M.R. Winchester, D.C. Duckworth and R.K. Marcus, in Glow Discharges Spectroscopies, Ed by R.K. Marcus. Plenum Press, New York, ch 7 (1990).
- C. Venzago, L. Ohanessian-Pierrard, M. Kasik, U. Collisi and S. Baude, J. Anal. Atom. Spectrom. 13, 189 (1998). https://doi.org/10.1039/a706758e
- N.E. Sanderson, E. Hall, J. Clark, P. Charalambous and D. Hall, Mikrochim. Acta (Wien) 1, 275 (1987). https://doi.org/10.1007/BF01199503
- J.W. Coburn and E. Kay, Appl. Phys. Lett. 19, 350 (1971). https://doi.org/10.1063/1.1653948
- D.M.P. Milton and R.C. Hutton, Spectrochim. Acta B 48, 1237 (1993). https://doi.org/10.1016/0584-8547(93)80006-G
- W. Schelles, S. De Gendt and R.E. Van Grieken, J. Anal. At. Spectrom. 11, 937 (1996). https://doi.org/10.1039/ja9961100937
- S.L. Tong and W.W. Harrison, Spectrochim. Acta B 48, 1237 (1993). https://doi.org/10.1016/0584-8547(93)80108-7
- S. De Gent, W. Schelles, R. Van Grieken and V. Muller, J. Anal. At. Spectrom. 10, 681 (1994). https://doi.org/10.1039/JA9951000681
- J.W. Coburn and E. Kay, Appl. Phys. Lett. 19, 350 (1970). https://doi.org/10.1063/1.1653948
- D.L. Donohue and W.W. Harrison, Anal. Chem. 47, 1528 (1975). https://doi.org/10.1021/ac60359a029
- D.C. Duckworth and R.K. Marcus, Anal. Chem. 61, 1979 (1989). https://doi.org/10.1021/ac00192a020
- J.S. Becker, A.I. Sprykin and H.-J. Dietze, Int. J. Mass Spectrom. Ion Processes 164, 81 (1997). https://doi.org/10.1016/S0168-1176(97)00021-9
- R.K. Marcus, J. Anal. At. Spectrom. 11, 821 (1996). https://doi.org/10.1039/ja9961100821
- R.K. Marcus, T.R. Harville, Y. Mei and C.R. Shick, Anal. Chem 66, 902A (1994).
- S. De Gendt, R. Van Grieken, S.K. Ohorodnik and W.W. Harrison, Anal. Chem. 67, 1026 (1995). https://doi.org/10.1021/ac00102a002
- R.C. Eanes and R.K. Marcus, Spectrochim. Acta B 55, 403 (2000). https://doi.org/10.1016/S0584-8547(00)00170-1
- R.K. Marcus, P.R. Cable, D.C. Duckworth, M.V. Buchanan, J.M. Pochkowski and R.R. Weller, Appl. Spectrosc. 46, 1992 (1992). https://doi.org/10.1366/0003702924123683
- D.P. Myers, M.J. Heintz, P.P. Mahoney, G. Pi and G.M. Hieftje, Appl. Spectrosc. 48, 1337 (1994). https://doi.org/10.1366/0003702944028029
- D.C. Duckworth, D.L. Donohue, S.H. Smith, T.A. Lewis and R.K. Marcus, Anal. Chem. 65, 2478 (1993). https://doi.org/10.1021/ac00066a013
- A.I. Sprykin, F.G. Melchers, J.S. Becker and H.J. Dietze, Fresenius J. Anal. Chem. 363, 570 (1995). https://doi.org/10.1007/BF00321325
- A. Bengston, Glow discharge plasmas in analytical spectroscopy. John Wiley & Sons, p. 141 (2003).
- R. Dorka, V. Hoffmann and M. Kunstàr, Presented at the European Winter Conference on Plasma Spectrometry, Hafjell, Norway (2001).
- J.I. Garcia-Alonso, D. Thoby-Schultzendorff, B. Giovannone and L. Koch, J. Anal. At. Spectrom. 8, 673 (1993). https://doi.org/10.1039/ja9930800673
- J.S. Crain, L.L. Smith, J.S. Yaeger and J.A. Alvarado, J. Radioanal. Nucl. Chem. 194, 133 (1995). https://doi.org/10.1007/BF02037619
- M.R. Smith, E.J. Wyse and D.W. Koppenaal, J. Radioanal. Nucl. Chem. 160, 341 (1992). https://doi.org/10.1007/BF02037109
- R.R. Ross, J.R. Noyse and M.M. Lardy, Radioact. Radiochem. 4, 24 (1993).
- J.J. Stoffel, J.F. Wacker, J.M. Bond, R.A. Kiddy and F.P. Brauer, Appl. Spectrosc. 48, 1326, (1994). https://doi.org/10.1366/0003702944028092
- M. Betti, G. Rasmunssen, T. Hiernaut, L. Koch, D.P.M. Milton and R.C. Hutton, J. Anal. At. Spectrom. 9, 385 (1994). https://doi.org/10.1039/ja9940900385
- K. Robinson and E.F.H. Hall, J. Met. 39, 14 (1987).
- M. Betti, G. Rasmunssen and L. Koch, Fresenius J. Anal. Chem. 355, 808 (1996).
- M. Betti, J. Anal. At. Spectrom. 11, 855 (1996). https://doi.org/10.1039/ja9961100855
- J.C. Velazco, J.H. Kolts and J.W. Setser, J. Chem. Phys. 69, 4357 (1978). https://doi.org/10.1063/1.436447
- L. Aldave de las Heras, F. Bocci, M. Betti and L.O. Actis-Dato, Fresenius J. Anal. Chem. 368, 95 (2000). https://doi.org/10.1007/s002160000529
- D.C. Duckworth, C.M. Barschick and D.H. Smith, J. Anal. At. Spectrom. 48, 875 (1993). https://doi.org/10.1039/ja9930800875
- M. Betti, S. Giannarelli, T. Hiernaut, G. Rasmussen and L. Koch, Fresenius J. Anal. Chem. 355, 642 (1996).
- L. Aldave de las Heras, E. Hrnecek, O. Bildstein and M.Betti. J. Anal. At. Spectrom. 17, 1011 (2002). https://doi.org/10.1039/B202450K
- M. Betti, G. Rasmussen and L. Koch, Fresenius J. Anal. Chem. 355, 808 (1996).
- World Nuclear Industry Handbook. Nuclear Engineering International (1999).
- D. Pecheur, J. Godlewski, P. Billot and J. Thomazet, Zirconium in the Nuclear Industry, Eleventh International Symposium ASTM STP 1295, ASTM, Philadelphia, PA, 94, 113 (1996).
- C. Lemaignan and T. Motta, in Materials Science and Technology Vol. 10B, Ed by B.R.T. Frost. VCH, Weinheim, Germany (1994).
- I.L. Branwell, P.D. Parson and D.R. Tice, Zirconium in the Nuclear Industry, Ninth International Symposium ASTM STP 1132, ASTM, Philadelphia, PA, 628, 642 (1991).
- L.O. Actis-Dato, L. Aldave de las Heras, M. Betti, E.H. Toscano, F. Miserque and T. Gouder, J. Anal. At. Spectrom. 15, 1479 (2000). https://doi.org/10.1039/b005668p
- M. Betti, “Analysis of samples of nuclear concern with Glow Discharge Atomic Spectrometry”, in Glow Discharges Plasmas in Analytical Spectroscopy, Ed by R.K. Marcus and J.A.C. Broekaert. John Wiley & Sons, p. 273 (2003).