Sara Castiglioni,* Ettore Zuccato, Chiara Chiabrando, Roberto Fanelli and Renzo Bagnati
Department of Environmental Health Sciences, Mario Negri Institute for Pharmacological Research, via La Masa 19, 20156 Milan, Italy. E-mail: [email protected]
Introduction
In recent years, the environmental fate of therapeutic drugs used by humans and animals (environmental pharmacology) has increasingly become recognised worldwide as an emerging issue. After administration, drugs can be excreted in high percentages as the parent compound and/or active metabolites in urine and faeces and reach wastewater treatment plants (WWTPs) and then be released into surface waters, soil and sediments.
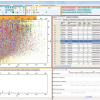
Concentrations of therapeutic drugs and their metabolites in waste and surface water are measured by highly selective and sensitive mass spectrometric techniques, such as gas chromatography-mass spectrometry (GC-MS) or high performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS). Using data obtained in this way, information about human metabolism of single active principles, and WWTPs characteristics (e.g., flow rate, population served), it is possible to calculate the environmental loads of therapeutic drugs and correlate them to their effective use (known from sales or production data).
Recently, this approach was used to study the environmental diffusion of illicit drugs and to estimate their effective use. The global production of major illicit drugs is comparable to that of widely used pharmaceuticals, and similar concentrations and behaviour are expected in WWTPs and in the environment. An initial study on cocaine showed that it was possible, from measurements of the main metabolite, benzoylecgonine, to estimate the number of doses that were used by the population served by a particular treatment plant. Other illicit drugs and related substances (amphetamines, morphine, heroin, cannabinoids, methadone) are now under investigation, and this method could be applied to integrate information obtained by conventional approaches, such as population surveys, consumer interviews and crime statistics.
We will consider here the analytical aspects involved in the measurements of illicit drugs and their metabolites in wastewater samples by HPLC-MS/MS.