John Hammond
Starna Scientific Ltd, 52–54 Fowler Road, Hainault Business Park, Hainault, Essex, IG6 3UT, UK
The summer of 2009 has been notable for two metrologically significant events: the annual meeting of the ISO REMCO Committee and the 12th BERM Symposium, neither of which has ever been held before in the UK. It is a massive credit to LGC that they were able to host both meetings and succeeded in making the BERM Meeting one of the best ever!
In this column John Hammond, the UK Industry Delegate to ISO/REMCO, reports on the proceedings and decisions of the meeting. I’ll be reporting on BERM 12 in the next column and also explaining the relationship between the UK Reference Materials Working Group, the BSI and ISO/REMCO. — Peter Jenks
Introduction
The 32nd meeting of ISO/REMCO was held from 3 to 6 July 2009, in Teddington, UK, and was hosted by LGC on behalf of the British Standards Institute (BSI). ISO/REMCO now has a membership of 70 members of the International Organization for Standardization (ISO) and liaison with 18 international organisations and six ISO internal committees.
Whilst, the liaisons are very important for the design and implementation of ISO/REMCO’s work programme, ISO/REMCO increases its activities to identify and involve additional experts, besides the REMCO delegates from ISO member bodies and liaisons, for its working groups developing guidance documents on specific RM issues. Most of the documents from ISO/REMCO directly affect work in other (international) organisations, and it is important to avoid duplication of effort, and where possible to share resources, namely the limited number of experts.
One of the main tasks of ISO/REMCO is preparing guidelines, for the preparation, characterisation, certification and use of reference materials and the competence assessment of reference material producers. These current (and proposed) guides and their relationships are shown in Figure 1. ISO/REMCO has updated its work programme during the meeting in Teddington. Main activities for 2009 and beyond include:
- a complete revision of the vocabulary related to reference materials (ISO Guide 30, 1992/Amd 1:2008), by extensive consultation with other liaison organisations.
- a complete revision of the guide on accompanying documentation (ISO Guide 31, 2000), expanded to cover all documentation for reference materials.
- a complete revision of the guide on the use of reference materials (ISO Guide 33, 2000).
- development of a new guide for the production and use of in-house Quality Control (QC) reference materials (ISO Guide 80).
- development of a guidance document for the production of RMs for “qualitative analysis”—testing of normative properties.
- a study into the matter of “traceability”, in particular how the concept is expressed on CRM certificates and related communications.
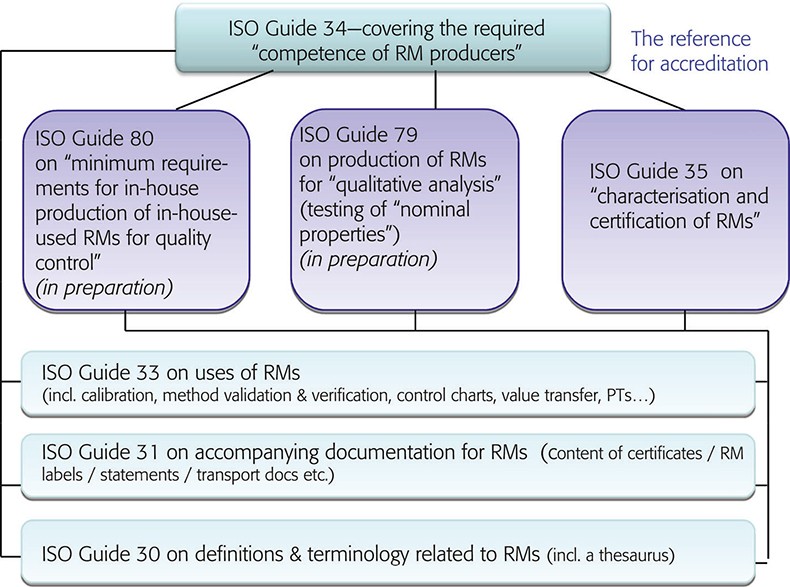
Figure 1. Structure of the ISO/REMCO Guides.
The technical report on characterisation (ISO/TR 10989:2009) has been published. A first draft, of ISO/TR 11773 on transportation issues was discussed at the meeting, and it is envisaged that a voting draft will be available for review at the next meeting. At present, these subjects do not require normative guidance, but it is important to publish the information available on the subjects in the form of reports.
Quality assurance in RM production
ISO Guide 34, the guide describing the requirements for the quality management system and the technical competences of reference material producers (RMP), has been fully revised and passed the required ISO voting unanimously. This 3rd revision aligns the document with ISO/IEC 17025:2005. ISO Guide 34 has become the worldwide “standard” for RMP accreditation and was included into the Mutual Recognition Arrangement (MRA) of National Metrology Institutes (NMIs) and other signatories of this MRA. The arrangement refers directly to ISO Guides 34 and 35 with respect to the requirements for certified reference materials (CRM).
Vocabulary
ISO/REMCO’s vocabulary is in need of revision, and has to define clearly the nuances specific to the use of terminology when applied to RMs. Meanwhile, an amendment to the present edition of ISO Guide 30 has been published introducing the new ISO/REMCO definitions for RM and CRM also into this guide. But there are other terms and concepts, such as the ones on “qualitative” measurements, which need further explanation and harmonisation. ISO/REMCO is pleased to note that the revised International Vocabulary for Metrology (VIM) is now published as ISO Guide 99, and will consider it alongside with other reference documents in the revision of Guide 30. There is also the intention to include into the revised Guide 30 references to equivalent or similar terms for facilitating the common understanding on RM issues with specific application sectors, e.g. pharmaceutical industry or microbiology.
Uses of RMs/CRMs
ISO Guide 33 is also under revision. The current document deals with uses of CRMs, excluding calibration, which is covered by ISO Guide 32. As shown in the guide structure in Figure 1, the new ISO Guide 33 will provide a normative reference to ISO Guide 34 and will cover the various types of RM uses, specified for certified and non-certified RMs. Consequently calibration will be included, which will eventually lead to the withdrawal of ISO Guide 32.
In-house preparation and use of RMs for QC
Many reference materials, in particular those used for laboratory-internal quality control, are either prepared within the laboratory or by an organisations own “quality/metrology department” at the same location. Several requirements for these RMs may be somewhat less severe than for RMs, and in particular for CRMs, which have a wider range of intended uses and which are produced for worldwide distribution. Therefore, a separate guidance document was deemed to be required. The new ISO Guide 80 will offer approaches to produce in-house RMs which can be used in-house for quality control purposes. This will not include applications such as calibration or validation of trueness for measurement methods.
Introductory guide to ISO/REMCO
A revised “Introduction to ISO/REMCO” has been prepared. This booklet will shortly be available on the ISO/REMCO website (www.iso.org/remco) and will explain the mission and structure of ISO/REMCO, available documents and the system of guides. Moreover, it will provide information how to communicate with the committee.
RMs for “qualitative analysis”—testing of normative properties
RMs for qualitative analysis are growing in number and in importance. So far, guidance on the production and use of RMs has been mainly focused on RMs/CRMs for quantitative measurements. For the authentication of food (e.g. wine, honey), the fight against doping in sports, the control of pesticides and other residues in foodstuff, the identification of microorganisms, these RMs are very important. In chemical analysis many requests are directed to the identification of a substance, rather than determining its concentration. Therefore, Working Group 13 is now tasked with compiling a list of nominal properties with examples to be considered for a future ISO Guide 79 “Reference materials for qualitative analysis (testing of nominal properties)”. The diversity of issues associated with drafting such a document has prompted ISO/REMCO to request assistance from contributors which are not REMCO delegates but have expertise in particular fields important for the development of this guide.
Committee structure
ISO/REMCO is structured as shown in Figure 2. The organisation consists of Working Groups (WG) set up to implement a specific task, under two Steering Groups (SG1 and SG2). The Chairman’s Advisory Group (CAG) provides input to the Chair from the Group convenors.
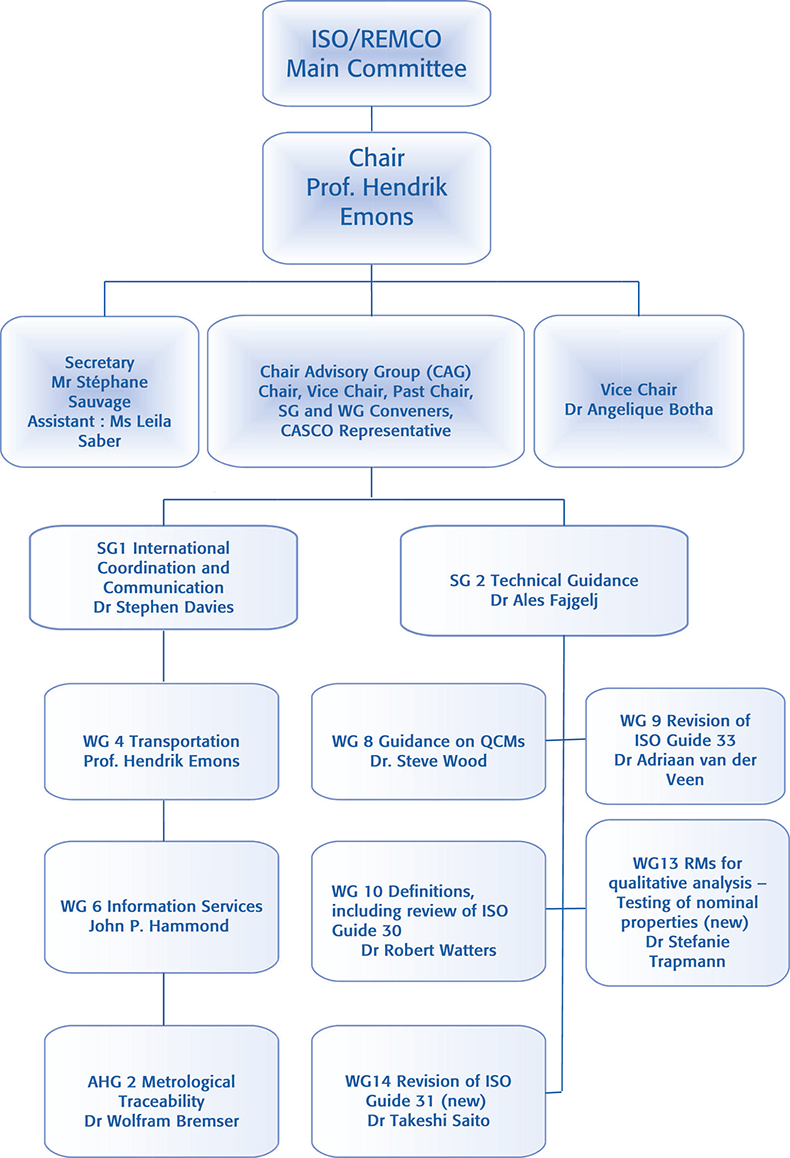
Figure 2.
Should you wish to contribute to the work of ISO/REMCO, please contact Prof. Dr Hendrik Emons ([email protected]) detailing your specific area of expertise.
Next meeting
The next ISO/REMCO meeting will be held in Hangzhou, China, from 3 to 7 May 2010.

