C. Burgessa and J.P. Hammondb
aBurgess Analytical Consultancy Limited, ‘Rose Rae’, The Lendings, Startforth, Barnard Castle, Co. Durham, DL12 9AB, UK
bStarna Scientific Ltd, 52–54 Fowler Road, Hainault Business Park, Hainault, Essex, IG6 3UT, UK
Background
Reviewing published documentation provides some interesting information as to how the requirement for the “calibration” of spectrometers has changed and evolved over approximately the last 40 years, and not least, the associated terminology of the science.
Why “40 years”? Because in 1976, the FDA promulgated a series of proposed GLP regulations, which were finalised under 21 Code of Federal Regulations (CFR) 58 in 1978 as well as the established cGMPs in 21 Code of Federal Regulations (CFR) 210 & 211. Purely by coincidence around this time, we began our own spectroscopic journeys performing measurements on old manual prism-based spectrometers such as the Beckman DU, Unicam SP 500 and Hilger Uvispek Mk IX. In those days, the focus was simply on technical performance characteristics.
By the late 1990s, in addition to the publication of guidance documents in key aspects of regulatory control, a defined qualification process was documented by various interested parties, including the current authors in several collaborative ventures.
These documents1,2 established the qualification framework as shown in Figure 1 and the recommended approach to UV/vis spectrometry.
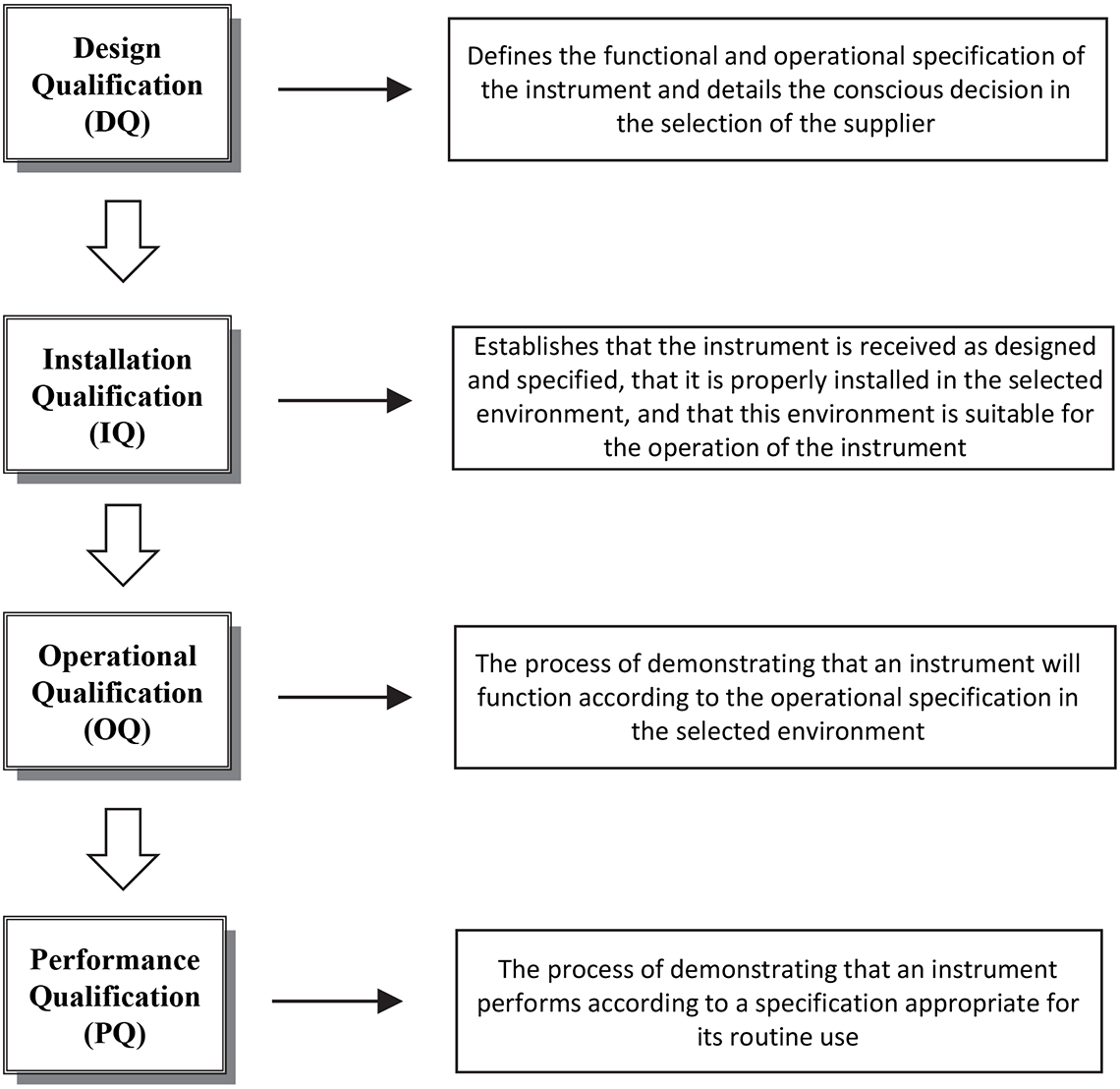
Figure 1. Qualification framework 1997–2000 Valid Analytical Measurement (VAM) Programme.
At the same time that the 4Qs qualification protocol was being developed, the central role of this essential qualification process was summarised by an instrument vendor, as shown in Figure 2.
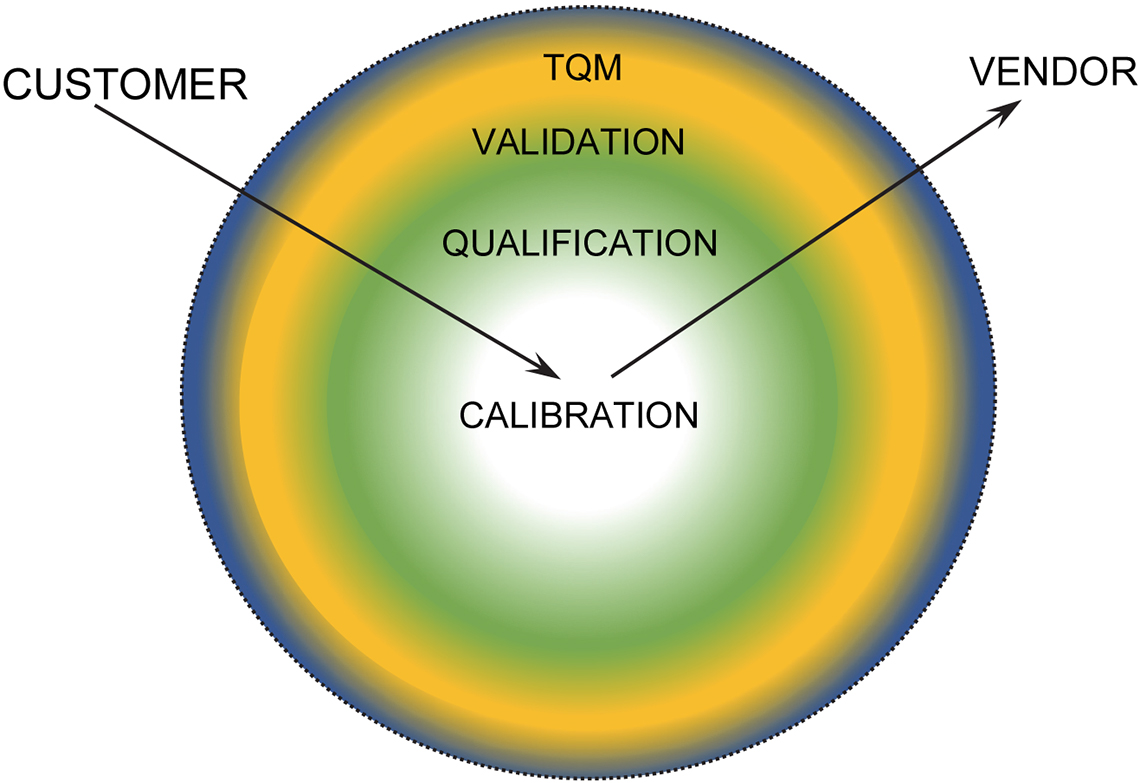
Figure 2. Vendor-derived validation/qualification and calibration (VQC) “Shells” circa 2000.
The statement made at that time was that:
The value of the chemical measurement depends upon the degree of confidence that can be placed on the result and thereby its “fitness for purpose”. If you couple this statement with any of the internationally recognised Quality Standards, one irrefutable observation is that both have a common requirement—effective equipment performance verification, often simply referred to as calibration. This requirement can be graphically shown as part of a series of concentric Validation/Qualification/Calibration (VQC) “shells”.
This structure affects both user and vendor, and the sequential process shown in Figure 2 will depend upon one’s initial starting position. As a user, the overall perspective is planned before specific tasks are undertaken. As an instrument manufacturer, clearly establishing calibration to specification is the first quality requirement of a newly produced instrument.
From the user’s viewpoint in practical terms this meant:
- Establishing total quality management (TQM) protocols.
- Formulating a validation plan.
- Qualifying the instrument or system.
- Ensuring initial (and maintaining) calibration.
From the vendor’s perspective this required:
- Ensuring calibration to specification.
- Assisting the end user in the qualification at the system location.
- Assisting/advising on additional validation & calibration/TQM aspects.
At the same time, the Royal Society of Chemistry’s Analytical Methods Committee, Instrumental Criteria Subcommittee published a detailed proposal on the selection of UV/vis/NIR systems.3
This concept is still valid today, some 20 years on, but as our title suggests—as always, there may be another and possibly better way.
Quality by design
At the same time as these fundamental validation, qualification and calibration principles were being defined and structured, in a parallel development, quality by design (QbD) as a concept was outlined by quality expert Joseph M. Juran in many publications, most notably Juran on Quality by Design.4
Designing for quality and innovation is one of the three universal processes of the Juran Trilogy, in which Juran describes what is required to achieve breakthroughs in new products, services and processes.5 Juran believed that quality could be planned, and that most quality crises and problems relate to the way in which quality was planned.
While QbD principles have been used to advance product and process quality in industry, and particularly the automotive industry, they have also being adopted by the pharmaceutical industry. As has been discussed many times in the column, international harmonisation is an on-going process, and QbD is no exception. In this case, regulators in the European Union (the European Medicines Agency), Japan and the US Food and Drug Administration (FDA) have furthered QbD objectives through the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH). ICH guidelines Q8 (on Pharmaceutical Development),6 Q9 (on Quality Risk Management)7 and Q10 (on Pharmaceutical Quality System)8 provide guidance for manufacturers to implement QbD into their own operations.
There is not the space within this article to expand further on this well-reviewed and discussed topic, but a simple search will reveal an extensive library devoted to this subject. However, the concept is introduced here because this guidance is being developed further into risk-based approaches to the management of quality, and are very much seen as the way forward in to the future.
Therefore, let us take a conscious decision to combine these two concepts and see what is produced. That is to say, if we bring this VQC shell structure up to date and apply a similar approach using QbD terminology, what is the result?
The result is shown in Figure 3.
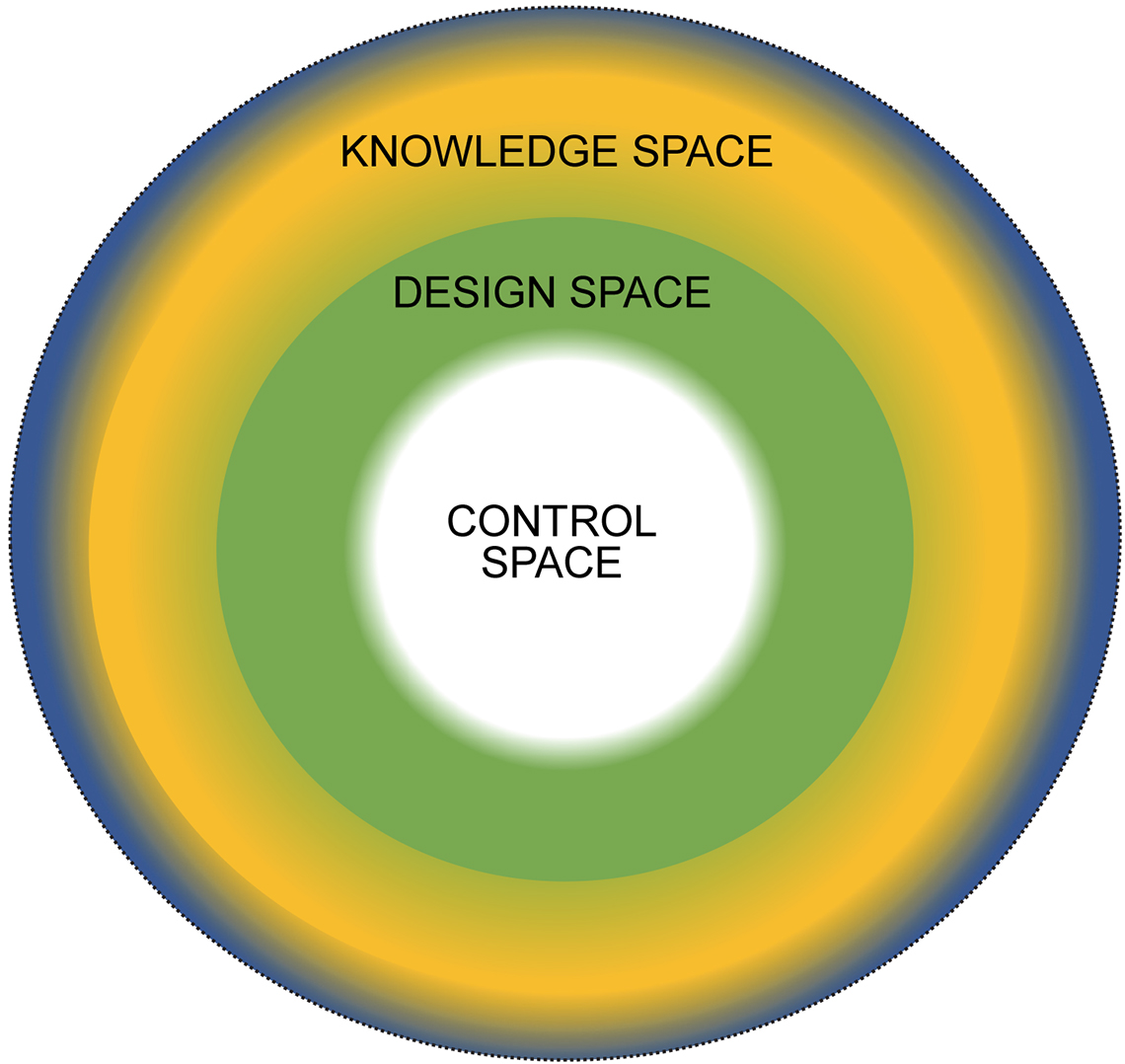
Figure 3. Qualification in a QbD environment.
If you compare Figure 2 and Figure 3 it is apparent that:
- Knowledge space and validation both form the outer shells.
- Design space links to qualification, and interestingly, both form the essential core framework on which the structure is built.
and
- Control space maps to calibration, the working centre of both structures.
If we now focus on Figure 3, this can be considered on the paper in two dimensions, i.e. a circle as a “space” in which you have:
- Knowledge space
- Design space
- Control space
But in practice, as discussed below, these spaces are in fact multi-dimensional surfaces encompassing the critical process parameters. Obviously, as with all concepts, these may be considered generic terms, and therefore may defined as such, but such definitions are often difficult, as by design, they will reflect the environment for which they are intended, e.g. mathematical, pharmaceutical etc. Some basic definitions are given from a spectroscopic viewpoint below.
Knowledge space
The theory and science associated with UV/vis spectroscopy, which after 70 years as an instrumental technique, is not inconsiderable and its critical process parameters (CPPs) are well established. In addition, the desired metrological outputs are specified as critical quality attributes (CQA).
Design space
A multivariate mathematical model relating the input CPPs and output CQAs to establish a region where at a level of probability the measurement process delivers “fitness for purpose” outcomes. Working within the design space is not considered as a change. Movement out of the design space is a change and would normally initiate a regulatory post approval change process. Design space is proposed by the applicant and is subject to regulatory assessment and approval.6
This is essentially the space in which a UV/vis spectrometer has been specified by the vendor and used for operational qualification (OQ).
Control space(s)
A more constrained region within the design space, sometimes called the “normal operating range”, is based on in-house specifications. This space covers the user-defined operational parameter range over which the instrument is going to be routinely used. For different applications, there may be different control spaces with the same overall design space. For example, the same spectrophotometric system may be used for one very specific application, over a defined wavelength and absorbance/transmittance range, or the system may be used for multiple applications, as shown in Figure 4.
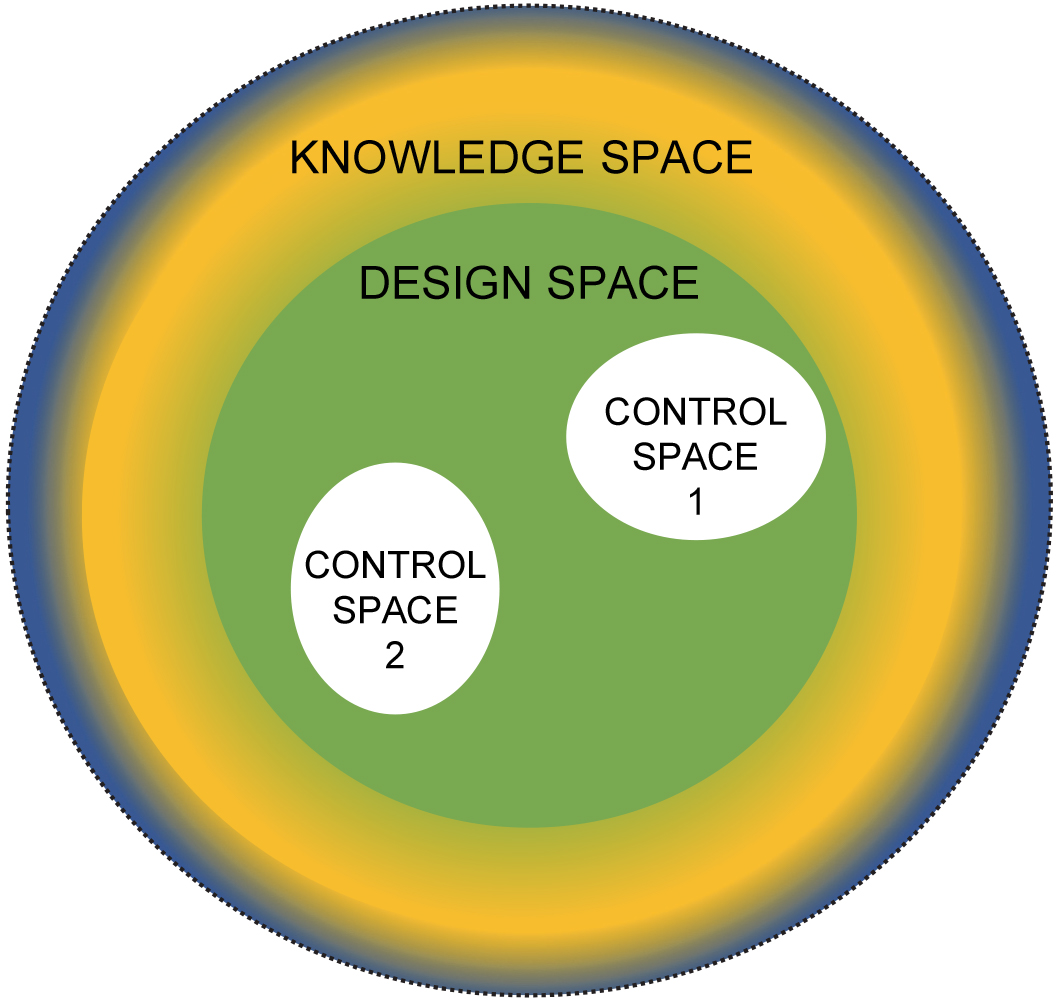
Figure 4. Multi application control spaces.
A CPP from a spectroscopic viewpoint is a physical optical characteristic that should be within an appropriate limit, range or distribution to ensure the desired metrological qualities (CQAs), and therefore in the case of UV/vis spectrometry, such CPPs are likely to include:
- Operational ranges of absorbance and wavelength.
- Wavelength accuracy and precision over the operational ranges.
- Photometric accuracy and precision over the operational ranges.
- Stray light.
- Spectral bandwidth.
So in a practice sense, we would suggest that with respect to the requirements nothing has changed, there is still the requirement to “calibrate” a spectro(photo)meter and it has always been there.
However, with these QbD concepts in place, the environment is more defined, is more structured and, once understood, easier to control. The next step is to introduce the “analysis of risk” into the process—but that is a topic for another day.
To bring the discussion “full circle”, also in 1975, Klaus Mielenz of NBS (now NIST) published a short paper on “The Nomenclature of Spectrometry”.9 Recent discussions have shown that it would appear we are no further forward in deciding whether the correct terminology for an instrument measuring transmittance across a defined wavelength range is an absorption spectrometer, or spectrophotometer; but again, this is probably a topic for another day?
References
- P. Bedson and M. Sargent, “The development and application of guidance on equipment qualification of analytical instruments”, J. Accred. Qual. Assur. 1, 265–274 (1996). https://doi.org/10.1007/s007690050083
- Guidance on Equipment Qualification of Analytical Instruments: UV-Visible Spectro(photo)meters. National Measurement System 2000–2003 Valid Analytical Measurement (VAM) Programme, Version 1.0, September 2000. https://doi.org/10.1039/a909502k
- “Report by the Analytical Methods Committee. Evaluation of analytical instrumentation Part XIII. Instrumentation for UV–visible–NIR spectrometry”, Analyst 125, 367–374 (2000). https://doi.org/10.1039/A909502K
- J.M. Juran, Juran on Quality by Design: The New Steps for Planning Quality into Goods and Services. Free Press (1992).
- J.M. Juran, “The quality trilogy: a universal approach to managing for quality”, Qual. Progr. 19(18), 19–24 (1986).
- ICH Q8 (R2), Pharmaceutical Development (August 2009).
- ICH Q9, Quality Risk Management (November 2005).
- ICH Q10, Pharmaceutical Quality Systems (June 2008).
- K.D. Mielenz, “The nomenclature of spectrometry”, Anal. Chem. 48, 1093 (1976). https://doi.org/10.1021/ac60371a011

